Professor Wei Zhu has published a paper in Signal Transduction and Targeted Therapy (STTT), a Nature Portfolio journal: Macrophage Metabolic Reprogramming Tackles the Challenge of Diabetic Chronic Wound Healing.
Diabetes mellitus is a major global public health concern. Diabetic foot ulcer (DFU), its most severe chronic complication, is the leading cause of amputation and mortality in diabetic patients. Persistent hyperglycemia triggers vascular dysfunction, peripheral neuropathy, and immune imbalance, resulting in impaired re-epithelialization, high infection risk, delayed healing, and high recurrence rate of wounds, which bring enormous suffering and medical burden to patients.
Macrophages are the core regulators of innate immunity and the key hub of wound healing. During normal wound healing, macrophages dynamically switch their phenotypes: in the early inflammatory phase, they are dominated by the M1 phenotype, which is responsible for clearing pathogens and initiating inflammation; in the resolution phase, they polarize to the M2 repair phenotype, which dominates tissue regeneration, collagen deposition, and angiogenesis. This phenotypic switch is metabolically regulated: M1 macrophages rely on glycolysis for energy supply, while M2 macrophages are highly dependent on mitochondrial oxidative phosphorylation (OXPHOS).
However, in the hyperglycemic microenvironment of diabetes, insulin resistance disrupts the glucose metabolic homeostasis of macrophages, blocks the transition from M1 to M2 phenotype, and keeps macrophages in a long-term pro-inflammatory M1 state, leading the wound to fall into a vicious cycle of chronic inflammation. Existing therapeutic strategies mostly focus on regulating the immune phenotype of macrophages, but fail to correct the metabolic disorder from the root cause, making it difficult to achieve long-term repair with limited clinical efficacy.
Vanadium, an essential trace element for the human body, has insulin-mimetic activity, and can precisely regulate glucose metabolism with immunomodulatory functions, making it an ideal target for resolving this metabolic disorder. However, its application in diabetic wound repair has not been systematically explored previously, and the challenges of controlled release of vanadium ions as well as dose-dependent systemic toxicity have severely restricted its clinical translation.
Building on these critical unmet clinical needs and core scientific challenges, Professor Wei Zhu from the School of Bioscience and Bioengineering, South China University of Technology (SCUT), in collaboration with Professor Gaoxing Luo and Researcher Yunlong Yu from the Institute of Burn Research of the Chinese People's Liberation Army (PLA), Southwest Hospital, Army Medical University, innovatively integrated vanadium with mesoporous bioactive glass (MBG). The team developed a V-MBG nanomaterial with dual functions of metabolic regulation and immunomodulation, and further constructed a matched glucose-responsive hydrogel delivery platform, which enables the on-demand release of vanadium ions in the wound microenvironment and realizes precise targeted therapy.
The relevant research findings, entitled Macrophage metabolic reprogramming by vanadium released from glucose-responsive bio-gel accelerates diabetic wound repair, were published online on April 23, 2026, in Signal Transduction and Targeted Therapy (STTT), a top-tier international medical journal with a latest impact factor (IF) of 52.7.
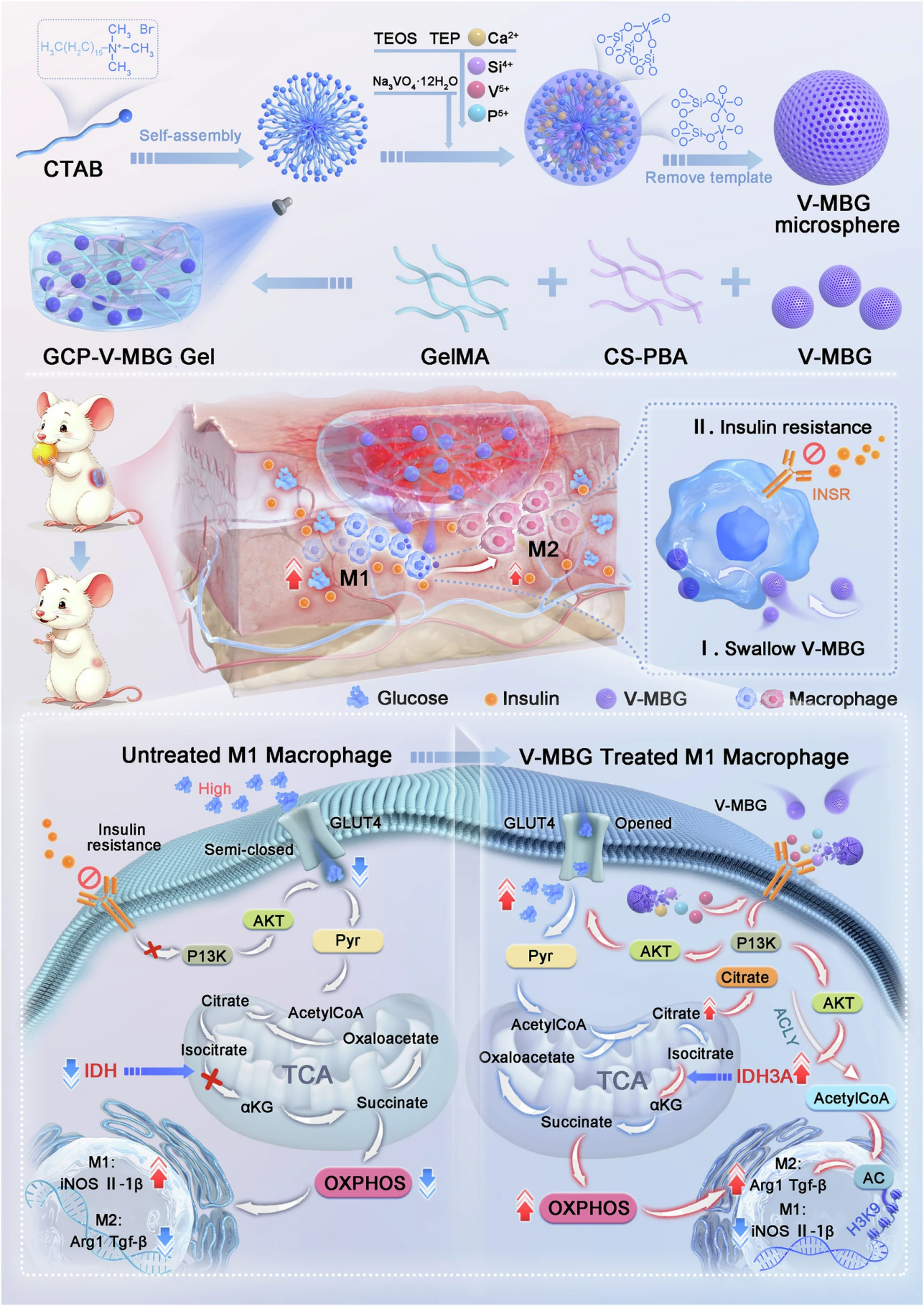
Figure 1. Synthesis of V-MBG nanospheres, regulatory mechanism of macrophage metabolism, and therapeutic application in diabetic wounds
This schematic fully presents the core design logic of the study, from the sol-gel synthesis route of V-MBG nanospheres, to the molecular mechanism of metabolic reprogramming and M2 polarization driven by activation of the INSR-PI3K signaling axis after phagocytosis by macrophages, and to the therapeutic application of the glucose-responsive hydrogel in diabetic wounds.
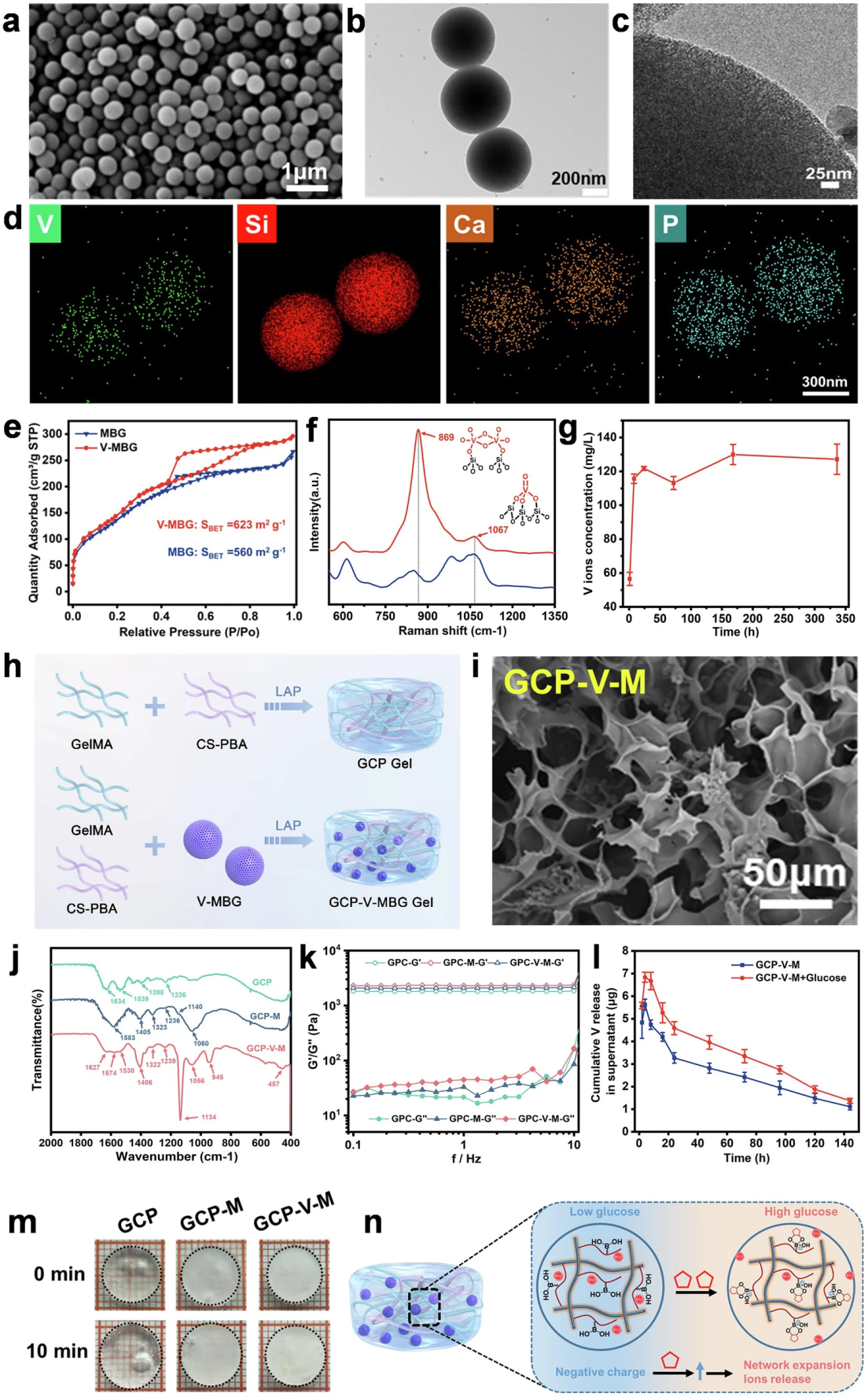
Figure 2. Physicochemical characterization of V-MBG nanospheres and GCP-V-MBG glucose-responsive hydrogel
The research team successfully synthesized V-MBG nanospheres based on the CaO-SiO2-P2O5 system via a modified sol-gel method using cetyltrimethylammonium bromide (CTAB) as the template. The nanospheres are spherical nanoparticles with uniform particle size (approximately 500±20.5 nm), with a well-ordered mesoporous structure and homogeneous distribution of vanadium, silicon, calcium, and phosphorus elements. The specific surface area is as high as 623 m2/g, enabling sustained and stable sustained release of vanadium ions in vitro.
To achieve targeted and controllable delivery of vanadium ions, the team grafted glucose-sensitive phenylboronic acid (PBA) onto carboxymethyl chitosan to prepare CS-PBA, which was co-assembled with gelatin methacrylate (GelMA) to construct a photo-crosslinked GCP hydrogel network. GCP-V-MBG (GCP-V-M) hydrogel was obtained after encapsulating V-MBG. Characterization results showed that the incorporation of V-MBG significantly improved the mechanical strength of the hydrogel without altering its three-dimensional porous morphology. In the high-glucose environment mimicking diabetic wounds, the hydrogel achieved concentration-dependent on-demand release of vanadium ions through the glucose-triggered matrix swelling effect, which not only ensured the local therapeutic concentration but also minimized the toxicity risk of systemic exposure, solving the core bottleneck for the clinical translation of vanadium-based preparations.
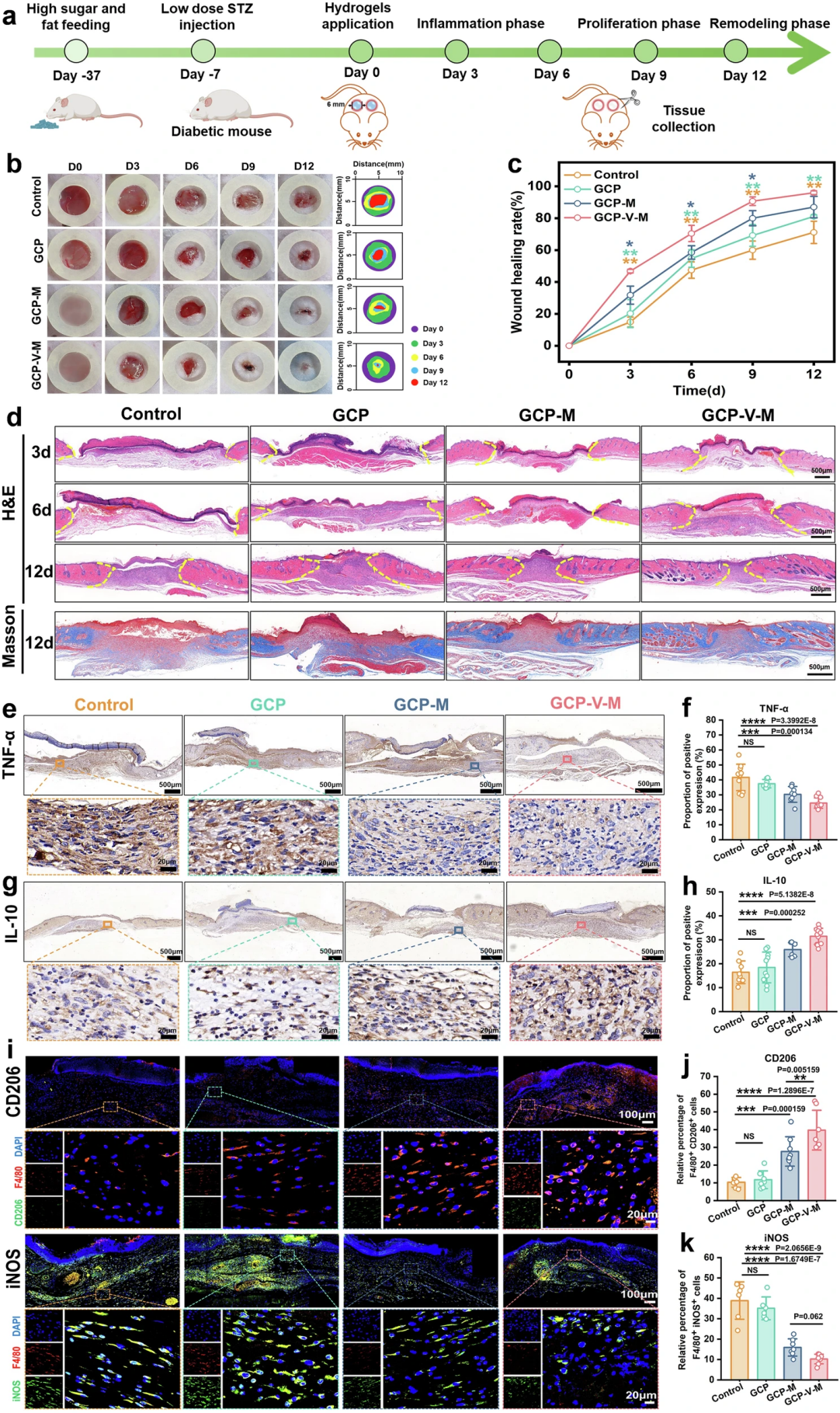
Figure 3. In vivo therapeutic efficacy and mechanistic validation of GCP-V-MBG hydrogel in promoting diabetic wound healing in mice
The research team successfully constructed a stable diabetic model in C57 mice through high-fat and high-sugar diet combined with low-dose streptozotocin (STZ) injection. Untargeted metabolomics confirmed the presence of significant metabolic disorders in the wound tissue of the mice, which perfectly mimicked the pathological characteristics of clinical diabetic wounds. The team established a 6 mm full-thickness skin defect model on the back of the mice, and divided the mice into four groups for treatment experiments: blank control group, GCP blank hydrogel group, GCP-MBG hydrogel group, and GCP-V-M hydrogel group.
The results showed that the GCP-V-M treatment group significantly accelerated wound closure at all time points, with a wound healing rate as high as 96.00±1.18% on day 12 of treatment, achieving nearly complete healing, and the therapeutic effect was significantly better than all control groups. Histological staining verified its excellent repair effect: H&E staining showed that the epidermis and granulation tissue layer were thicker in the GCP-V-M group on day 3 and day 6 of treatment, indicating that the early inflammatory response and regeneration process were effectively activated; on day 12 of treatment, the epidermis and granulation tissue layer were significantly thinner, indicating that the wound entered the mature remodeling stage faster. Masson's trichrome staining confirmed that the collagen deposition in the wound of the GCP-V-M treatment group was significantly increased, and the collagen arrangement was more regular, providing good structural support for wound healing.
Immunohistochemistry and immunofluorescence staining further analyzed the core therapeutic mechanism: GCP-V-M treatment significantly down-regulated the expression of pro-inflammatory factors TNF-α and IL-6 in the wound, and greatly up-regulated the expression of anti-inflammatory factor IL-10, effectively inhibiting excessive chronic inflammation in the wound. CD31 staining results confirmed that the hydrogel also significantly promoted the formation of new blood vessels in the wound, providing sufficient blood supply for tissue regeneration. The detection results for macrophages showed that on day 4 of treatment, the proportion of CD206-positive M2 macrophages in the wound of the GCP-V-M group was significantly increased, while the proportion of iNOS-positive M1 macrophages was significantly decreased, confirming that the hydrogel can efficiently drive the polarization of macrophages to the repair M2 phenotype in vivo, and reshape the anti-inflammatory and repair microenvironment of the wound from the root cause.
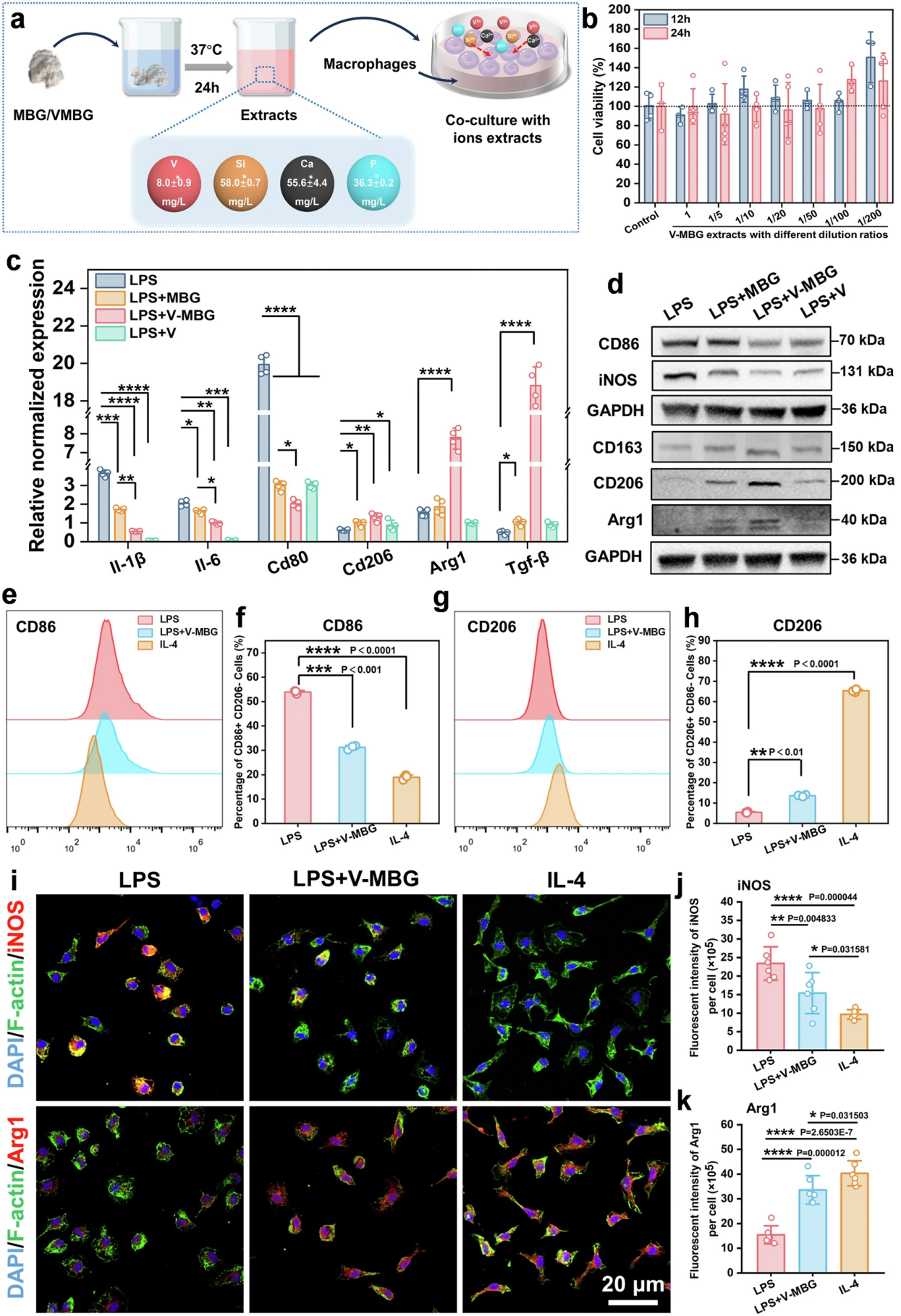
Figure 4. Direct in vitro validation of V-MBG-regulated macrophage polarization towards M2 phenotype
To clarify the direct effect of V-MBG on regulating macrophage polarization, the team conducted systematic in vitro cell experiments. ICP detection of the prepared V-MBG and MBG extracts confirmed that there was no significant difference in the concentrations of silicon, calcium, and phosphorus between the two groups, and vanadium ions were unique to V-MBG, excluding the interference of other ions. CCK-8 and live/dead staining experiments confirmed that the V-MBG extract had no obvious cytotoxicity at the experimental concentration, and could slightly improve macrophage viability at a specific dilution ratio, with good biocompatibility.
In the LPS-induced M1 macrophage model, RT-qPCR results showed that V-MBG treatment significantly down-regulated M1-related genes such as Il-1β, Il-6, and Cd80, and up-regulated M2-related genes such as Cd206, Arg1, and Tgf-β; Western blot further verified this result at the protein level, showing that V-MBG treatment significantly reduced M1 markers such as CD86 and iNOS, and increased the expression of M2 markers such as CD163, CD206, and Arg1. Flow cytometry and immunofluorescence staining results confirmed that V-MBG treatment significantly reduced the CD86+/CD206- M1 macrophage population and increased the CD206+/CD86- M2 macrophage population, with a pro-M2 polarization effect comparable to that of the classic M2 inducer IL-4, directly verifying the core role of V-MBG in driving macrophage phenotypic switch.
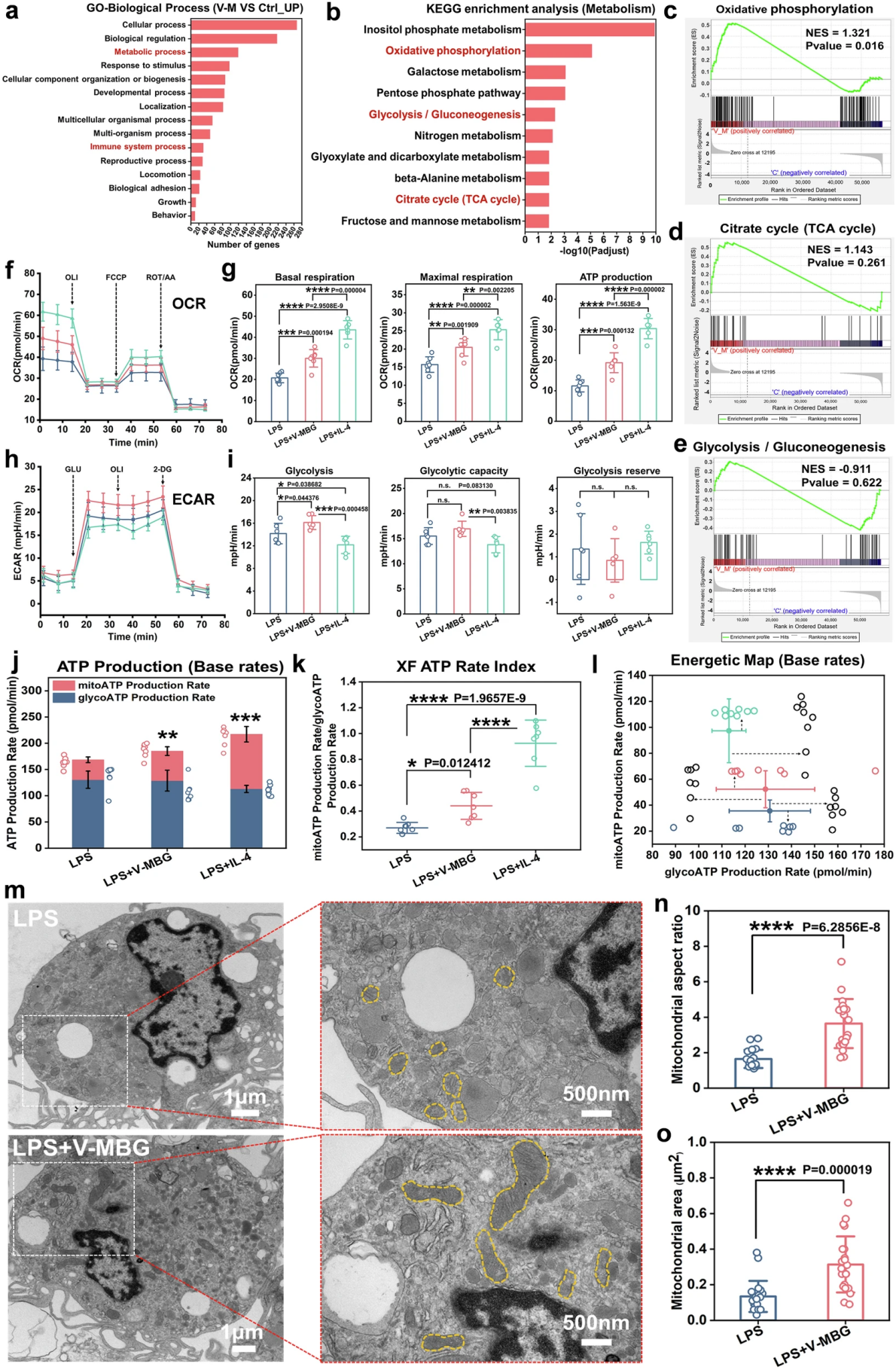
Figure 5. V-MBG remodels macrophage energy metabolism towards an OXPHOS-dominant phenotype
To reveal the core metabolic mechanism of V-MBG-driven macrophage polarization, the team conducted transcriptomics and functional metabolic experiments. Transcriptome sequencing results showed that the differentially expressed genes between macrophages in the V-MBG treatment group and the control group were significantly enriched in metabolic process and immune system process; KEGG enrichment analysis found that core energy metabolic pathways such as oxidative phosphorylation, glycolysis/gluconeogenesis, and tricarboxylic acid (TCA) cycle were significantly activated, suggesting that V-MBG drives phenotypic switch by remodeling macrophage energy metabolism.
Seahorse energy metabolism assay results showed that V-MBG treatment significantly increased the oxygen consumption rate (OCR) of macrophages, and the basal respiration, maximal respiratory capacity, and ATP production were greatly elevated, with the mitochondrial oxidative phosphorylation level comparable to that of the IL-4 treatment group; while the extracellular acidification rate (ECAR) assay showed that V-MBG only slightly increased glycolytic activity, far less than its promoting effect on mitochondrial respiration. ATP production rate analysis confirmed that the total ATP production in the V-MBG treatment group was significantly increased, mainly derived from OXPHOS, and macrophages completed the metabolic conversion to OXPHOS dominance. Transmission electron microscopy observation provided evidence at the structural level: after V-MBG treatment, the mitochondria in macrophages showed increased volume, enhanced fusion, and expanded membrane surface area, with significantly increased mitochondrial aspect ratio and area, providing a direct structural basis for the elevated OXPHOS level.
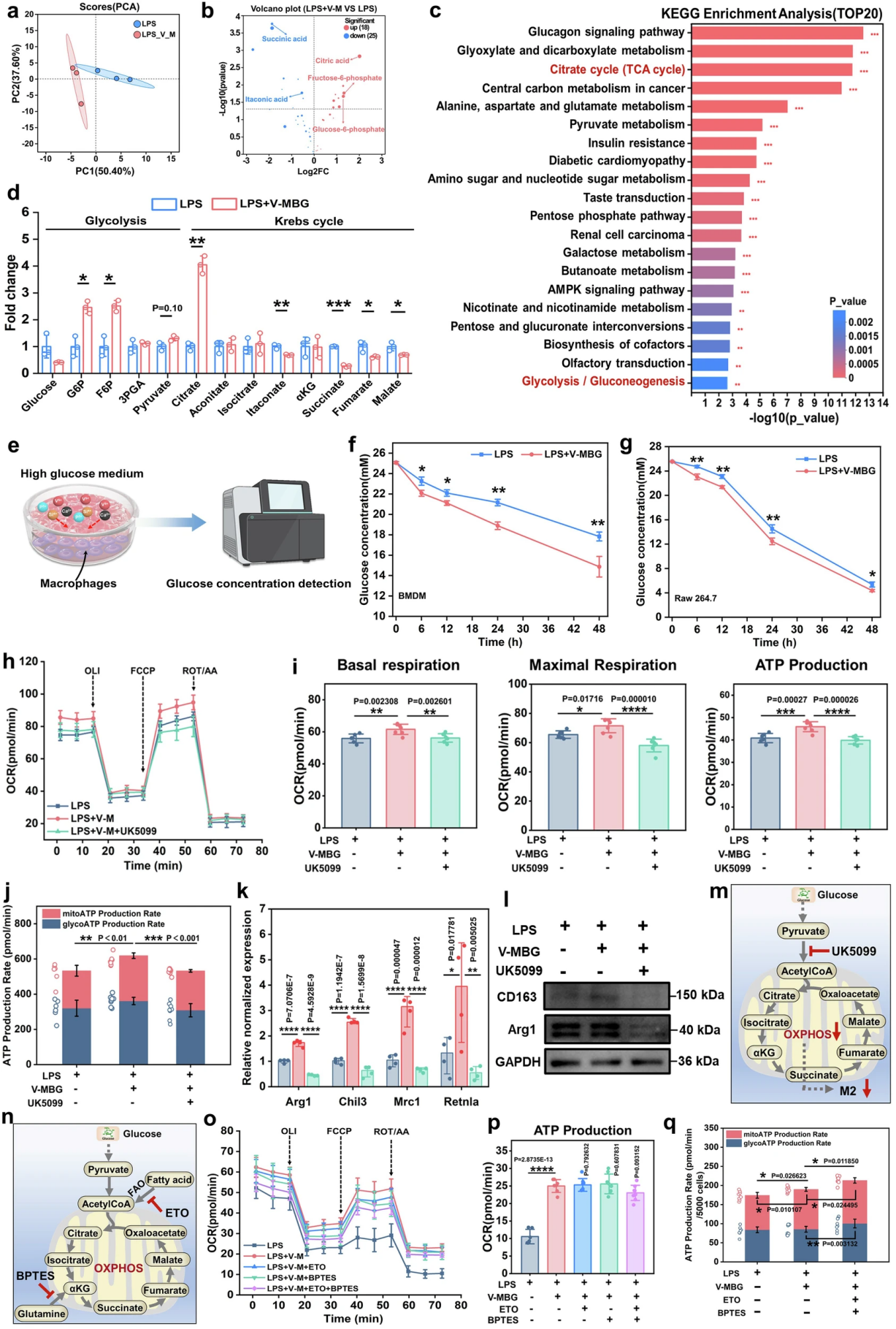
Figure 6. Glucose-driven mitochondrial OXPHOS is the core prerequisite for V-MBG-induced M2 polarization of macrophages
Through metabolomics and functional blocking experiments, the team finally confirmed that glucose metabolism is the core energy source for V-MBG to achieve macrophage metabolic reprogramming. Central carbon metabolomics analysis showed that there were significant differences in the metabolic profiles of macrophages between the V-MBG treatment group and the control group, with a total of 43 differential metabolites screened, mainly concentrated in the glycolysis and TCA cycle pathways. Quantitative results of metabolites showed that after V-MBG treatment, the levels of glycolysis intermediates G6P, F6P and the key TCA cycle metabolite citrate in macrophages were significantly increased, the levels of pro-inflammatory metabolites such as itaconate and succinate were decreased, while the intracellular glucose content was decreased, indicating a significant increase in glucose uptake and metabolic flux; time gradient detection also confirmed that V-MBG treatment significantly enhanced the glucose uptake and consumption capacity of macrophages.
Functional blocking experiments further verified its core necessity: after blocking mitochondrial pyruvate import with UK5099, the basal respiration, maximal respiration and ATP production of macrophages induced by V-MBG were significantly decreased, and OXPHOS-derived ATP was greatly reduced. Meanwhile, the expression of M2-related genes and proteins induced by V-MBG was significantly inhibited, confirming that mitochondrial pyruvate uptake is a key link for V-MBG to achieve metabolic reprogramming and M2 polarization. Inhibitor experiments confirmed that inhibition of fatty acid oxidation (ETO) and glutaminolysis (BPTES) only had a slight effect on mitochondrial respiration of macrophages, did not significantly reduce OXPHOS-derived ATP, and could not reverse V-MBG-induced M2 polarization. It was finally clarified that V-MBG mainly relies on glucose-driven mitochondrial OXPHOS as the core energy source to achieve M2 polarization of macrophages.
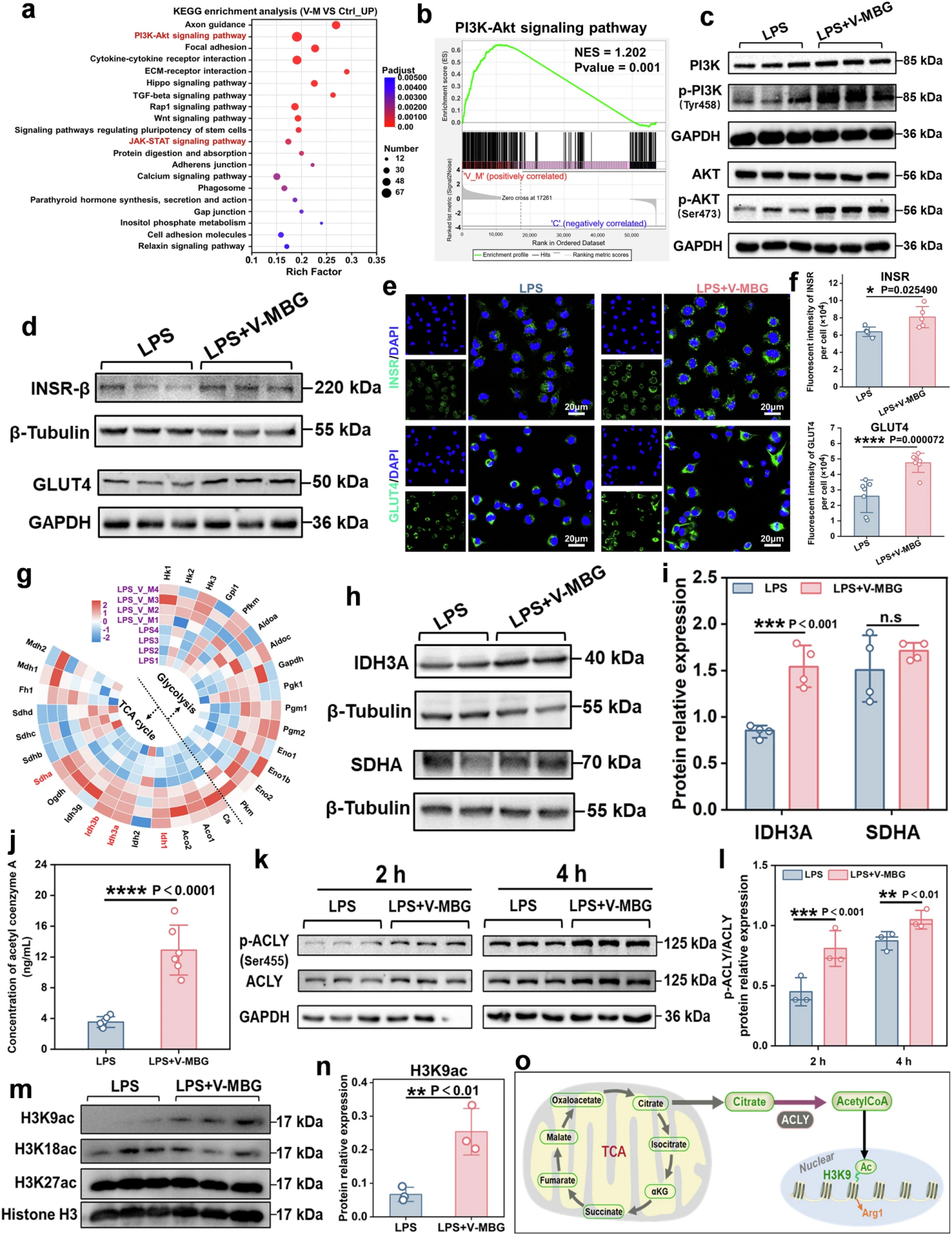
Figure 7. Molecular and epigenetic mechanisms of V-MBG-driven macrophage metabolic reprogramming and polarization
Through multi-omics and molecular experiments, the team further explored the upstream molecular regulatory network. Transcriptome KEGG enrichment analysis and GSEA verification showed that the PI3K-AKT pathway was significantly activated; Western blot confirmed that the phosphorylation levels of PI3K and AKT were significantly increased after V-MBG treatment, while the JAK-STAT pathway was not significantly activated, clarifying that PI3K-AKT is the core signaling axis for V-MBG to exert its effects.
Further studies found that V-MBG significantly up-regulated the gene and protein expression of insulin receptor (INSR), and significantly increased the expression of glucose transporter GLUT4, while GLUT1 expression had no significant change; subsequent inhibitor experiments confirmed that GLUT4 is the main transporter for V-MBG-induced glucose uptake in macrophages, and GLUT1 only plays a compensatory role, clarifying that V-MBG enhances glucose uptake in macrophages by activating the INSR-PI3K-AKT axis to up-regulate GLUT4 expression. Meanwhile, this signaling axis can restore the TCA cycle flux inhibited by LPS by up-regulating the expression of IDH3A, the key rate-limiting enzyme of the TCA cycle, providing sufficient metabolic support for OXPHOS.
In addition, the team also discovered a key epigenetic regulatory mechanism: the increased citrate level in macrophages after V-MBG treatment significantly elevated the content of acetyl-CoA (Ac-CoA) by activating ATP citrate lyase (ACLY), thereby up-regulating the acetylation level of histone H3K9ac. Through the citrate-Ac-CoA-H3K9ac epigenetic axis, it promotes the expression of M2-related genes, providing an amplification effect at the epigenetic level for macrophage phenotypic switch.
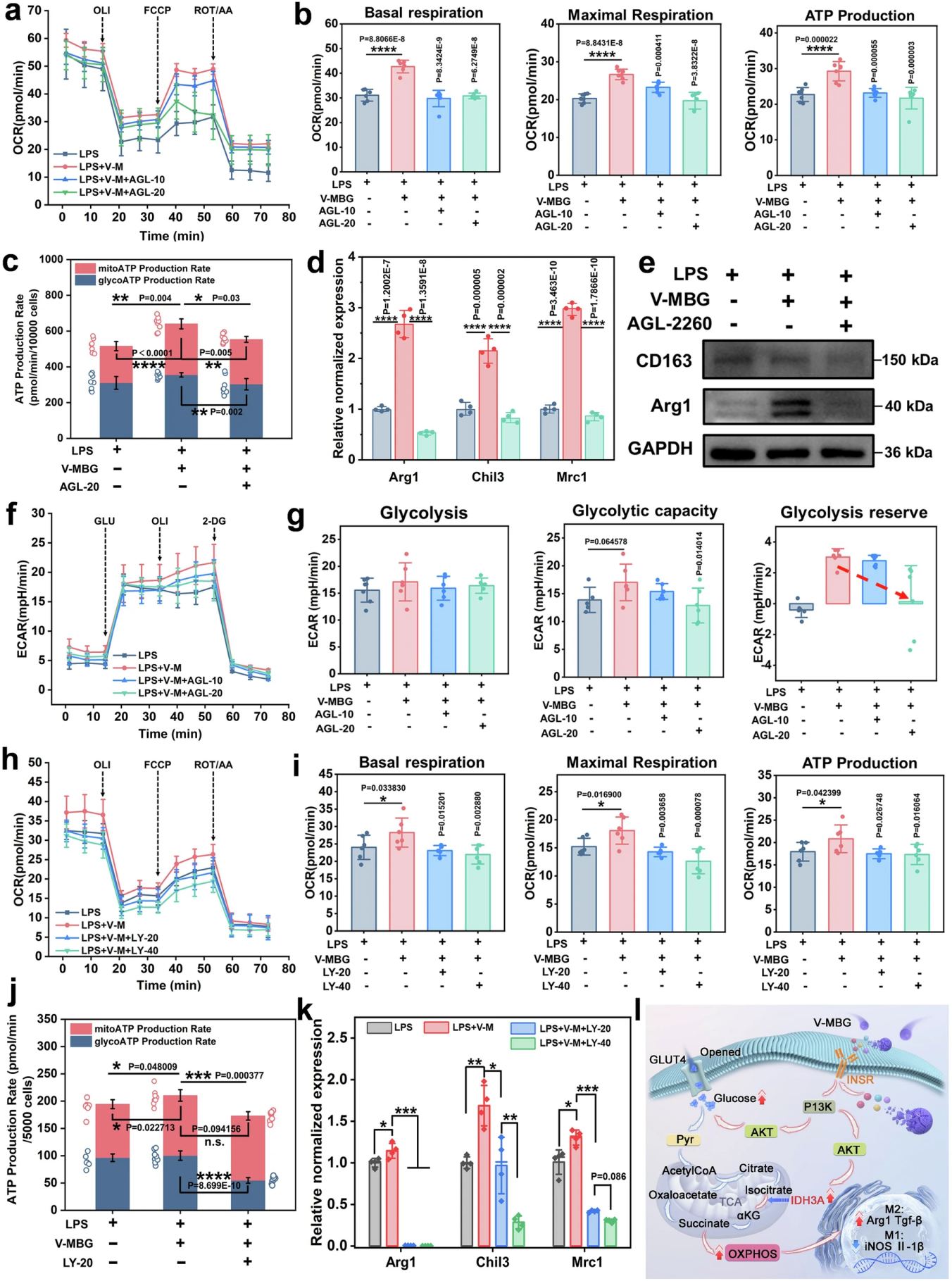
Figure 8. INSR-PI3K signaling axis mediates glucose-driven OXPHOS and M2 polarization of macrophages
Through systematic inhibitor rescue experiments, the team finally completed the closed-loop verification of the core pathway. After treatment with the INSR-specific inhibitor AGL-2263, the mitochondrial basal respiration, maximal respiration and ATP production of macrophages induced by V-MBG decreased in a dose-dependent manner, both OXPHOS-derived and glycolysis-derived ATP were significantly reduced, and the expression of M2-related genes and proteins induced by V-MBG was significantly inhibited, confirming that INSR is the upstream core target for V-MBG to exert metabolic regulation and pro-polarization effects.
After treatment with the PI3K inhibitor LY294002, the OCR of macrophages decreased in a dose-dependent manner, basal respiration, maximal respiration and ATP production were significantly reduced, glycolytic activity was also significantly inhibited, and the M2 polarization effect induced by V-MBG was also reversed in a dose-dependent manner. Treatment with the GLUT4-specific inhibitor fasentin significantly reduced the mitochondrial respiration level of macrophages and completely reversed the up-regulation of M2-related gene and protein expression induced by V-MBG.
Finally, the study fully confirmed the core mechanism of action: vanadium ions released from V-MBG can activate INSR in macrophages, thereby initiating the PI3K-AKT signaling axis. On the one hand, it promotes GLUT4-mediated glucose uptake, and on the other hand, it up-regulates IDH3A expression to enhance TCA cycle flux, which ultimately synergistically increases the level of glucose-driven mitochondrial OXPHOS. Meanwhile, the citrate-Ac-CoA-H3K9ac epigenetic axis further amplifies the M2 polarization effect, and finally realizes macrophage metabolic reprogramming and repair phenotype switch to accelerate diabetic wound healing.
Research Significance and Translation Prospects
This study is the first to systematically reveal the effect and complete mechanism of vanadium in promoting diabetic wound repair by targeting macrophage immuno-metabolism, breaking through the long-term bottleneck of existing diabetic wound treatment that focuses on immune phenotype regulation but neglects root correction of metabolism, and provides a novel immuno-metabolic regulation research paradigm for chronic wound treatment. The glucose-responsive GCP-V-M hydrogel developed by the team realizes on-demand sustained release of vanadium ions in the wound microenvironment, which significantly improves the therapeutic effect while minimizing the systemic exposure and toxicity risk of vanadium, solving the core pain point for the clinical translation of vanadium-based preparations.
Compared with FDA-approved stem cell and growth factor therapies, this hydrogel system is a cell-free, growth factor-free therapeutic strategy, which has significant advantages such as clear composition, good stability, ready-to-use, lower cost, and easy large-scale production, providing a new solution with great translation potential for the clinical treatment of diabetic foot ulcers. At the same time, the innovative concept proposed in this study of repairing chronic wounds through targeted immunometabolic reprogramming with biomaterials also provides new ideas and theoretical support for research in other inflammatory diseases, ischemic tissue injury and regenerative medicine fields.
This work was financially supported by the National Key R&D Program of China (2021YFA1101100), the National Natural Science Foundation of China (82202459, 82472570), Natural Science Foundation of Chongqing Municipality (CSTB2022NSCQ-MSX0153), Science and Technology Research Program of Chongqing Municipal Education Commission (KJZD-M202512806), General Program of China Postdoctoral Science Foundation (2023M734261), Special Funding for Postdoctoral Research Projects of Chongqing (2022CQBSHTB3043). Academic Talent Development Fund-Southwest Hospital Boqing Tuoju, China (2025BQTJ-1).