Professor Wei Zhu Publishes Review in ACS Nano Medicine: Red Blood Cell Engineering—From Traditional Carriers to Armored Modification
Recently, Professor Wei Zhu’s team at South China University of Technology, in collaboration with Professor Chunyan Yao’s team from Southwest Hospital of Army Medical University, published a review article entitled“Red Blood Cell Engineering: From Traditional Carriers to Armored Modification”in ACS Nano Medicine. The paper systematically outlines the developmental trajectory of red blood cell (RBC) engineering from conventional carrier-based applications to functional “armored” modification, comprehensively summarizing the field’s key technical strategies, major application scenarios, and future translational challenges. The work offers a new perspective for interdisciplinary research in transfusion medicine, drug delivery, and regenerative medicine.Dr. Yuxiang Wang, a postdoctoral researcher at the School of Biology and Biological Engineering, South China University of Technology, is the first author, and Zhenrui Xue from Southwest Hospital of Army Medical University is the co-first author. Professor Wei Zhu of South China University of Technology and Professor Chunyan Yao of Southwest Hospital of Army Medical University are the corresponding authors.
Article link:https://pubs.acs.org/doi/10.1021/acsnanomed.6c00023
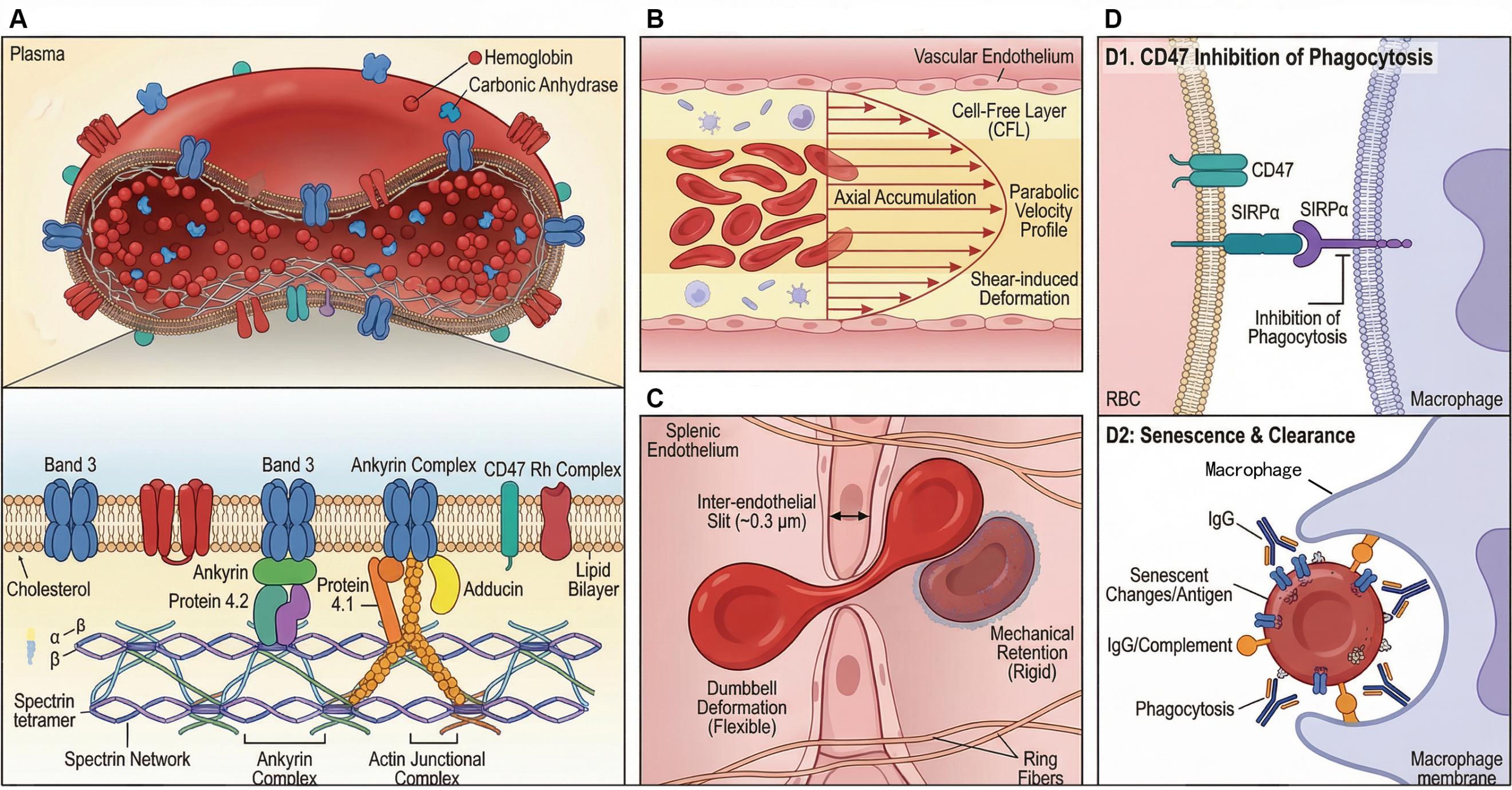
Figure 1.Structure, mechanical properties, and in vivo metabolic process of RBCs.
Why Has RBC Engineering Attracted So Much Attention?
RBCs are among the most abundant cells in the human body. They possess unique advantages, including broad availability, excellent natural biocompatibility, long circulation time, and relatively low immunogenicity. For these reasons, RBCs have long been regarded as ideal biological carriers. Whether serving as oxygen transporters in transfusion medicine or as platforms for drug delivery, immune regulation, and tissue repair, RBCs have demonstrated enormous application potential.
However, traditional RBC-based applications also face significant limitations. For example, RBCs are prone to membrane damage, morphological alterations, reduced deformability, and functional deterioration during storage and reinfusion. At the same time, relying solely on the intrinsic properties of native RBCs is often insufficient to meet the demands of complex disease treatment, which increasingly requires targeting capability, stability, and multifunctionality. Against this backdrop, RBC engineering has gradually evolved from simply “using RBCs as natural carriers” to “precisely and programmably modifying RBCs.”
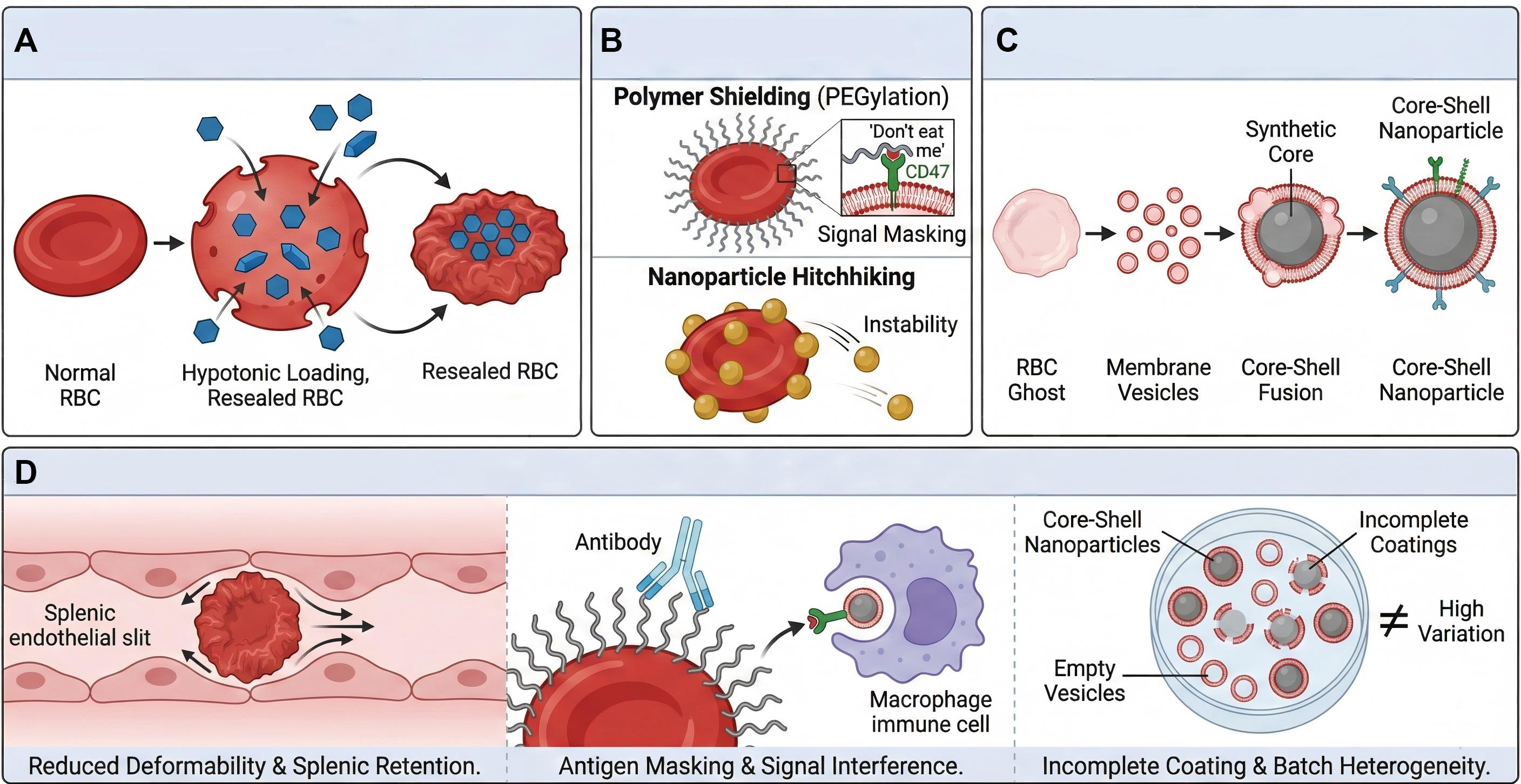
Figure 2. Strategies and limitations of traditional RBC engineering.
What Are “Armored RBCs”?
So-called armored RBCs refer to RBCs that are modified through material engineering, interface engineering, or biomimetic construction to form protective and functional composite structures on or around the cell surface, thereby enhancing their stability, tolerance, and performance in complex environments. Compared with conventional strategies that mainly rely on cargo loading or membrane camouflage, armored modification places greater emphasis on the coordinated regulation of RBC mechanical properties, surface chemical characteristics, and biological behavior.
Such strategies can not only protect RBCs, to some extent, from storage injury, cryodamage, and external stress, but can also endow them with new functions, such as improved circulatory stability, targeted delivery, and smart responsive behavior. More importantly, they may provide a novel bridge linking nanomedicine with living-cell therapies.
From Traditional Carriers to Armored Modification: The Evolution of RBC Engineering
The review points out that the development of RBC engineering has generally progressed through three stages: from the use of native RBCs, to external surface modification, and finally to intelligent armored engineering.
At the early stage, researchers mainly employed RBCs as natural biological carriers for drug encapsulation, enzyme delivery, or nanoparticle camouflage using RBC membranes. The major advantage of this stage lay in exploiting the excellent biocompatibility and long circulation properties of RBCs to improve the in vivo stability and safety of therapeutic agents.
Subsequently, with advances in nanomaterials, interface chemistry, and biofabrication, RBC engineering entered the stage of surface engineering. Through polymer modification, membrane functionalization, inorganic material coating, and biomimetic construction, RBCs were no longer merely “used,” but actively “reshaped.” In particular, the emergence of armored strategies has transformed RBCs from traditional transport tools into multifunctional living platforms with protective, sensing, responsive, and therapeutic potential.
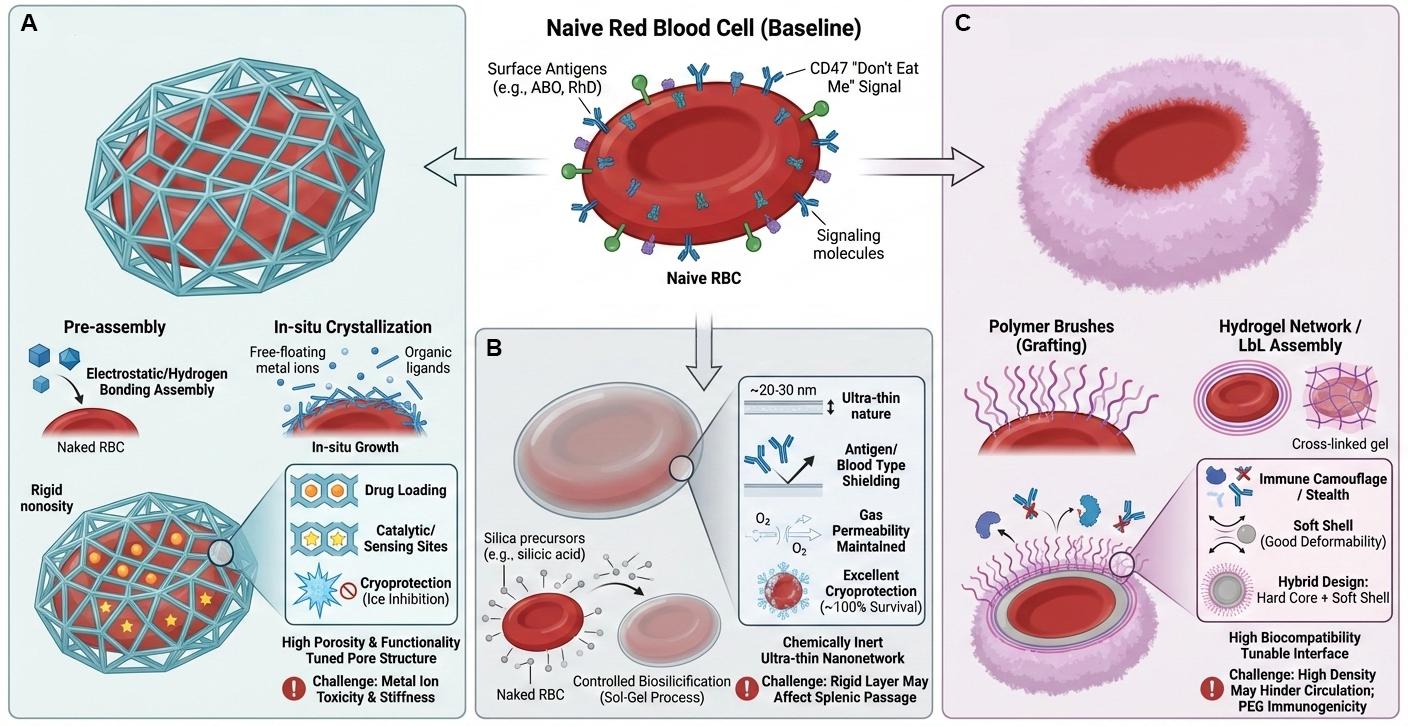
Figure 3. Three major strategies for constructing armored RBC exoskeletons and their representative characteristics.
Three Major Application Directions Show Broad Prospects
1. Transfusion Medicine and RBC Preservation
The quality of RBC storage has always been a critical issue in transfusion medicine. During storage, RBCs gradually undergo “storage lesions,” including membrane lipid remodeling, oxidative damage, and reduced deformability, which can impair their survival and function after reinfusion. Armored RBC strategies, by introducing protective shells, low-damage processing methods, and standardized preparation workflows, are expected to improve RBC stability during storage and recovery, thereby enhancing transfusion outcomes.
The review particularly emphasizes that one of the most important future tasks in this area is to establish dedicated critical quality attributes for armored RBCs, while also integrating closed, low-shear, and standardized processing workflows to promote the translation of these technologies from the laboratory to practical applications.
2. Drug Delivery and Disease Treatment
RBC engineering has also shown tremendous promise in drug delivery. Taking advantage of the natural long-circulation and immune-evasive properties of RBCs, researchers have used them to deliver small-molecule drugs, proteins, nucleic acids, and even nanoparticles, thereby improving therapeutic efficacy while reducing systemic toxicity. Armored modification further expands this capability by enabling RBCs to retain excellent biocompatibility while achieving higher structural stability and greater adaptability to challenging biological environments.
By coupling RBCs with functional materials, targeting molecules, and responsive elements, armored RBCs may become integrated platforms capable of transport, controlled release, and therapeutic regulation, with broad applications in cancer treatment, inflammation control, and precision medicine.
3. Regenerative Medicine and Living-Cell Therapeutic Platforms
Beyond transfusion and drug delivery, armored RBCs are also regarded as promising candidates for regenerative medicine. Compared with conventional nonliving materials, RBCs inherently possess flexibility, circulatory adaptability, and favorable safety profiles. Through mechanics-chemistry composite engineering, their functional boundaries can be further expanded. The review suggests that, in the future, by incorporating smart responsiveness, programmable regulation, and synthetic biology design, armored RBCs may serve as important intermediaries connecting nanomedicine with living-cell therapies, opening new avenues for tissue repair, microenvironment regulation, and the development of novel cell-based therapeutic products.
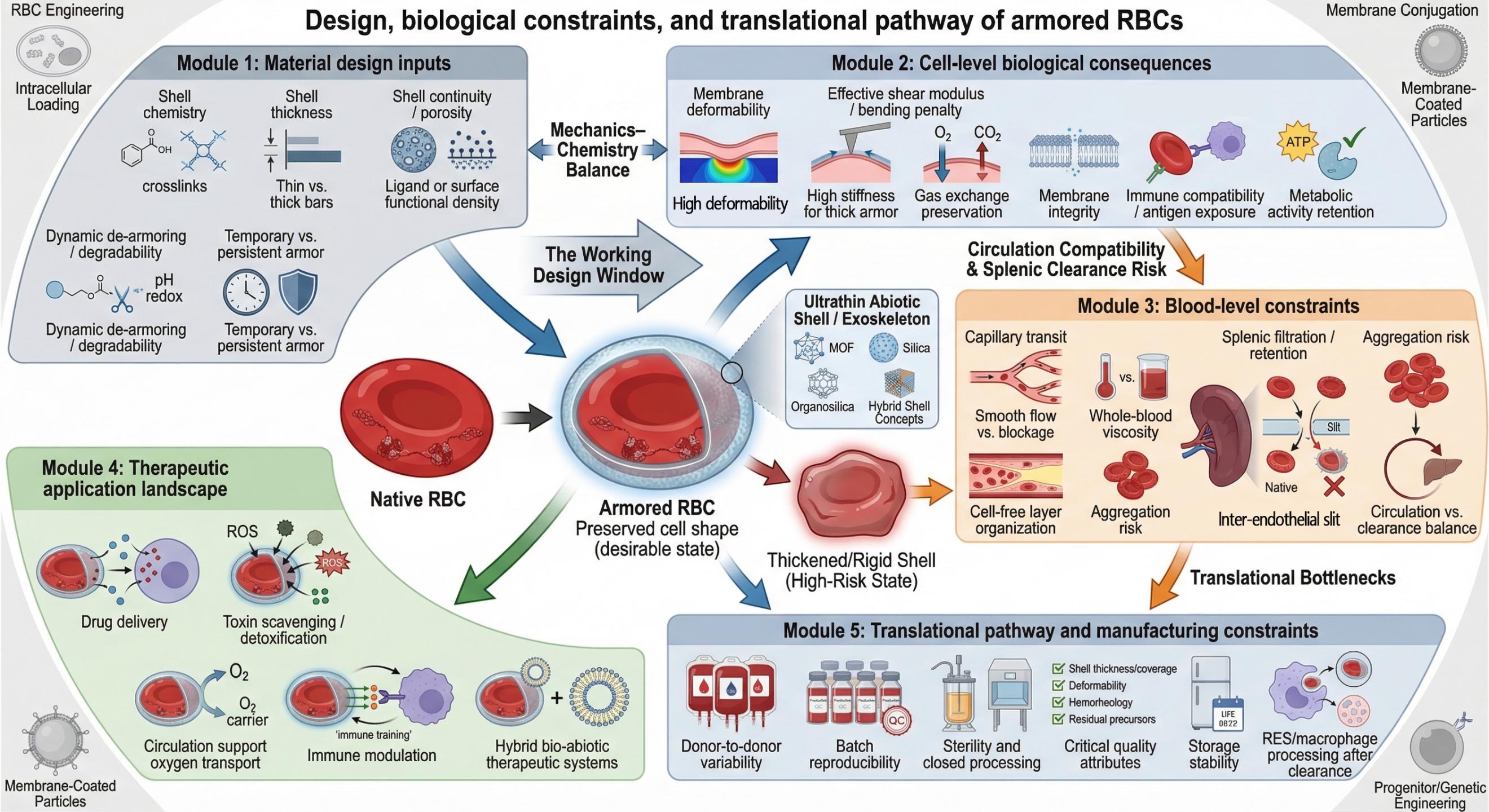
Figure 4. Design principles, biological constraints, and translational pathways of armored RBCs.
Broad Prospects, but Clinical Translation Still Faces Challenges
Although research on armored RBCs has made remarkable progress, a series of key issues must still be addressed before these technologies can truly enter clinical practice and large-scale application. First, different materials and modification strategies may alter the hemorheological behavior, membrane mechanical properties, and immune recognition characteristics of RBCs. How to strike a balance between enhanced functionality and physiological compatibility remains one of the central scientific questions in the field. Second, the long-term safety, degradation behavior, and biocompatibility of armored RBCs during storage, reinfusion, and in vivo metabolism still require more systematic evaluation.
In addition, donor variability, the lack of unified processing standards, and insufficient control of shear-induced damage during preparation all pose practical challenges to scale-up and clinical translation. The article notes that future progress in this field will depend not only on innovations in materials science, but also on deeper integration with hemorheology, immunology, and synthetic biology.
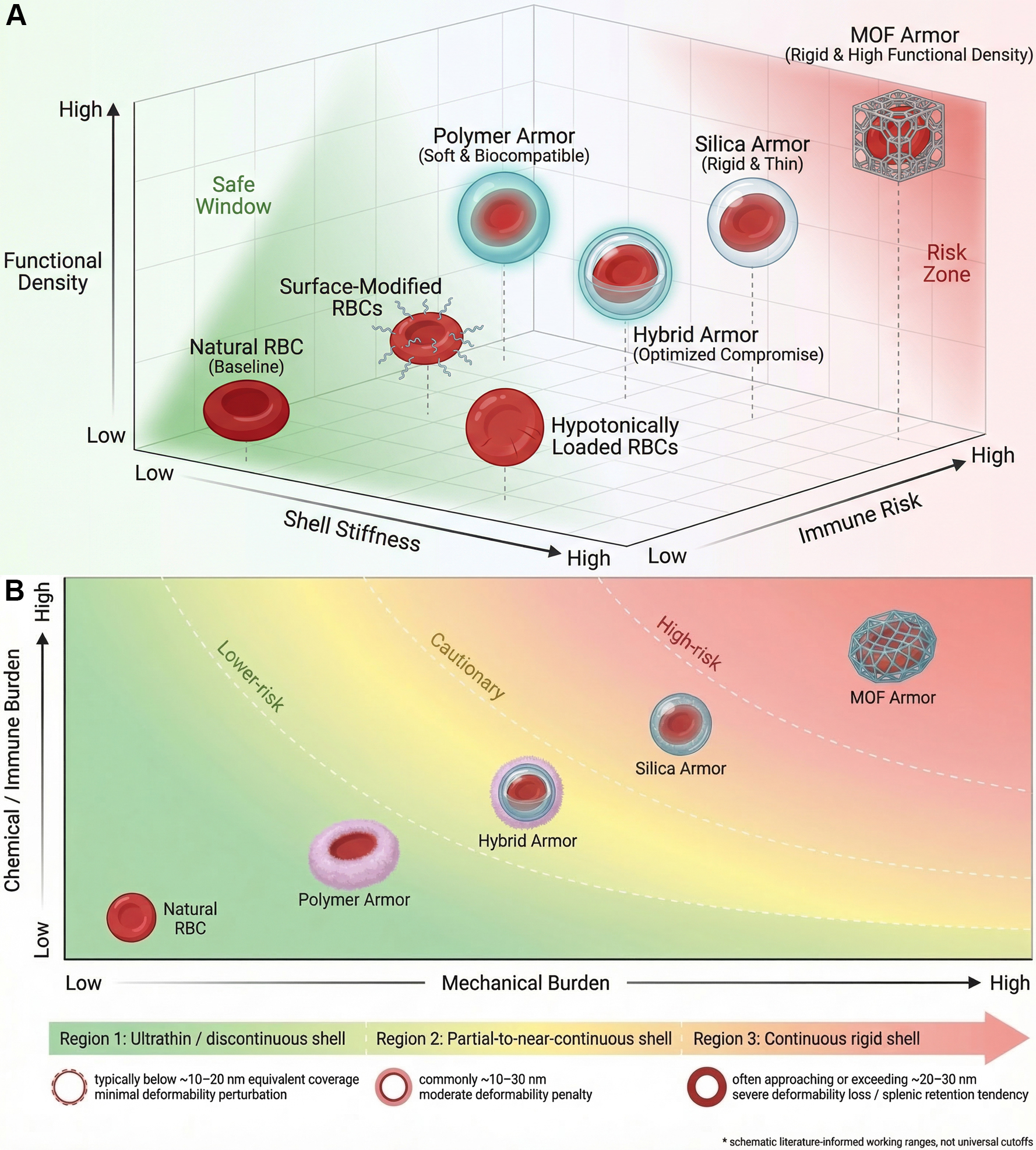
Figure 5. The multidimensional design space and current working boundaries of armored RBCs.
Multidisciplinary Integration Drives RBC Engineering into a New Stage
Overall, this review demonstrates that RBC engineering is moving beyond the traditional concept of a “biological carrier” toward a new stage of intelligent armored living platforms. By constructing composite systems that combine mechanical protection, interface regulation, and functional responsiveness, armored RBCs are expected to play increasingly diverse and far-reaching roles in transfusion medicine, drug delivery, and regenerative medicine.
In the future, with the gradual clarification of critical quality standards, continuous optimization of processing workflows, and the improvement of biosafety evaluation systems, armored RBCs are expected to become an important new platform linking nanomedicine and living cell therapy, injecting new momentum into precision medicine and the development of innovative biopharmaceutical technologies.
This work was supported by the National Natural Science Foundation of China(Grant Nos. 22372061 and 22572061), the Guangdong Science and Technology Program (Grant No. 2024B1111130002), the Guangzhou Science and Technology Plan Project (Grant No. 2024A03J0163), and the Fundamental Research Funds for the Central Universities.