Professor Wei Zhu Published Paper in PNAS: Oocyte-Inspired Universal Whole-Cell Vaccines Against Tumor Heterogeneity
Recently, Professor Wei Zhu from the School of Biology and Biological Engineering at South China University of Technology, in collaboration with Professor Jimin Guo from the College of Materials Science and Engineering at Beijing University of Chemical Technology and Professor C. Jeffrey Brinker from the University of New Mexico jointly published a research paper entitled "Oocyte Inspired Universal Whole Cell Vaccines Against Tumor Heterogeneity" online in Proceedings of the National Academy of Sciences (PNAS). Guo Sishi, a doctoral student from the School of Biological Sciences and Engineering at South China University of Technology.
Link to the paper:
https://doi.org/10.1073/pnas.2520276123
Drawing inspiration from the zona pellucida, a natural protective structure in oocytes, the research team developed biomimetic shells via three interface engineering schemes to biomimetic cell. This approach enabled whole-cell protein protection for whole tumor cell vaccines and allowed regulation of their mechanical properties at the single-cell level. The biomimetic structures yielded a dual breakthrough in "antigen integrity protection + immune efficiency enhancement," and established a novel paradigm combining individualization and universality for whole tumor cell vaccines, thereby providing an important translational solution for tumor immunotherapy and prevention.
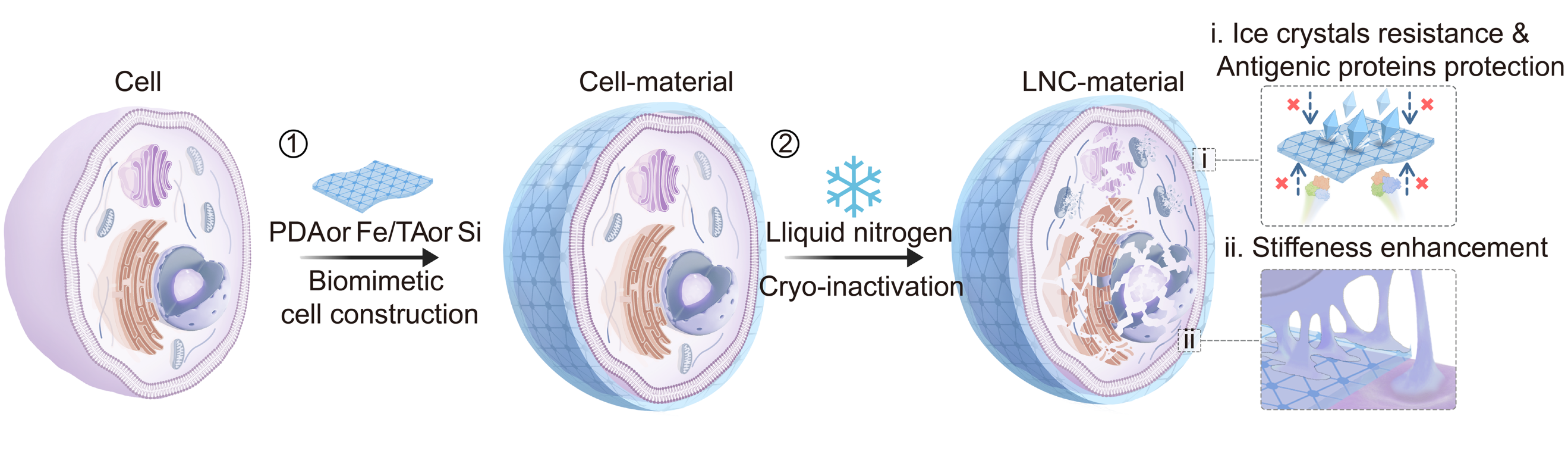
Figure 1. Construction of biomimetic cells.
The biomimetic shell protects proteins from mechanical damage induced by ice crystals during vaccine cryopreservation, thereby reducing the loss of immunogenic proteins and maintaining the abundance and diversity of whole-cell antigens. By modulating the mechanical properties of vaccine cells, the shell enables dendritic cells (DCs) to perceive the biomimetically enhanced mechanical stimulation. These mechanical signals are then converted into biochemical signals, activating cellular pathways and regulating DC functions, ultimately improving the efficiency of phagocytosis and antigen presentation of the vaccine cells.
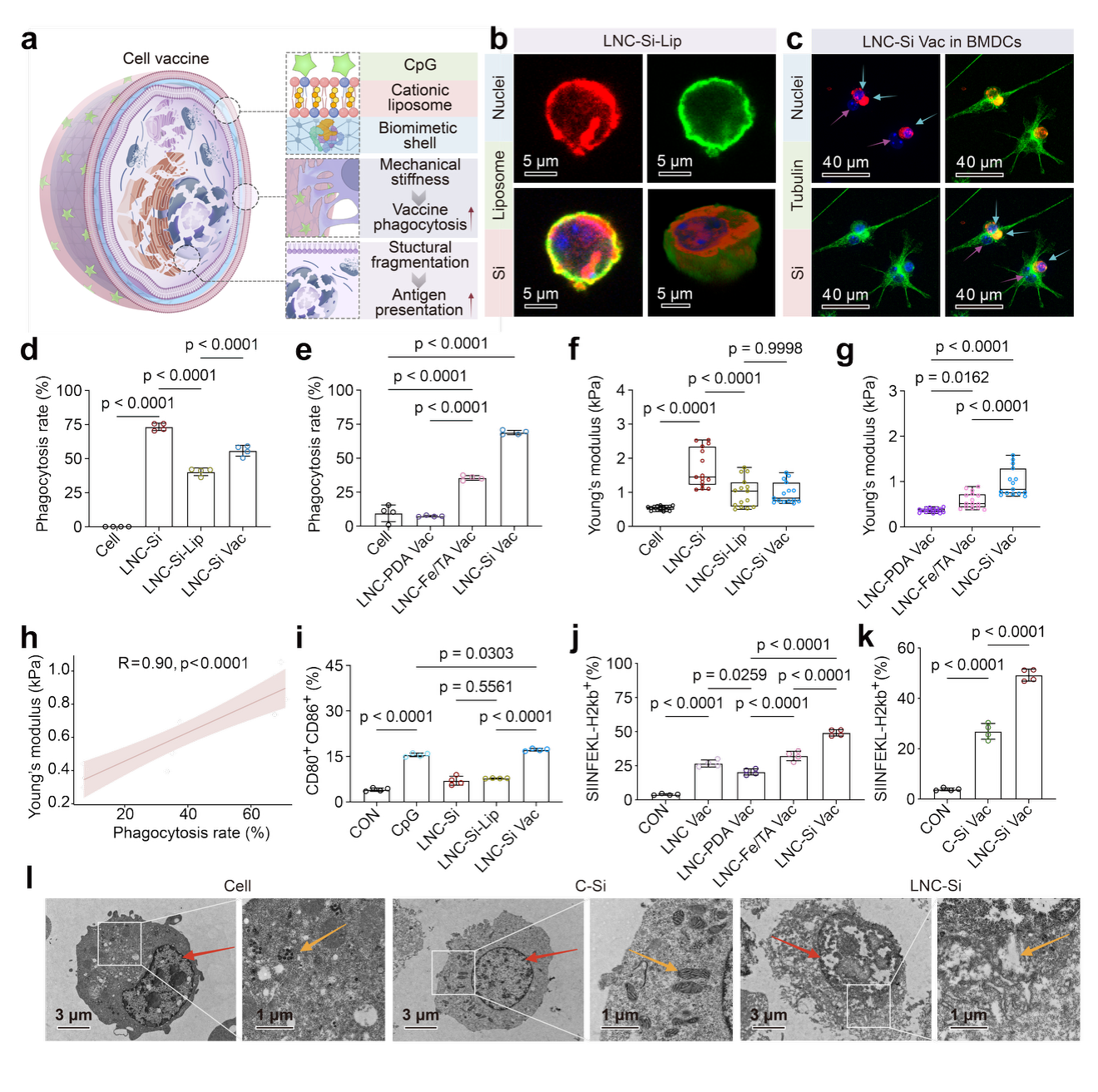
Figure 2. Formulation of vaccines based on biomimetic cells.
In a prophylactic model of aggressive B16-F10 mouse melanoma, vaccination with F10 Vac extended median survival by nearly 50% compared to controls. By day 12 post-challenge, the tumor volume in vaccinated mice was approximately one-sixth that of the control group, indicating strong prophylactic antitumor efficacy. In a therapeutic model, vaccination prolonged median survival by 28.1%, demonstrating a clear therapeutic benefit.
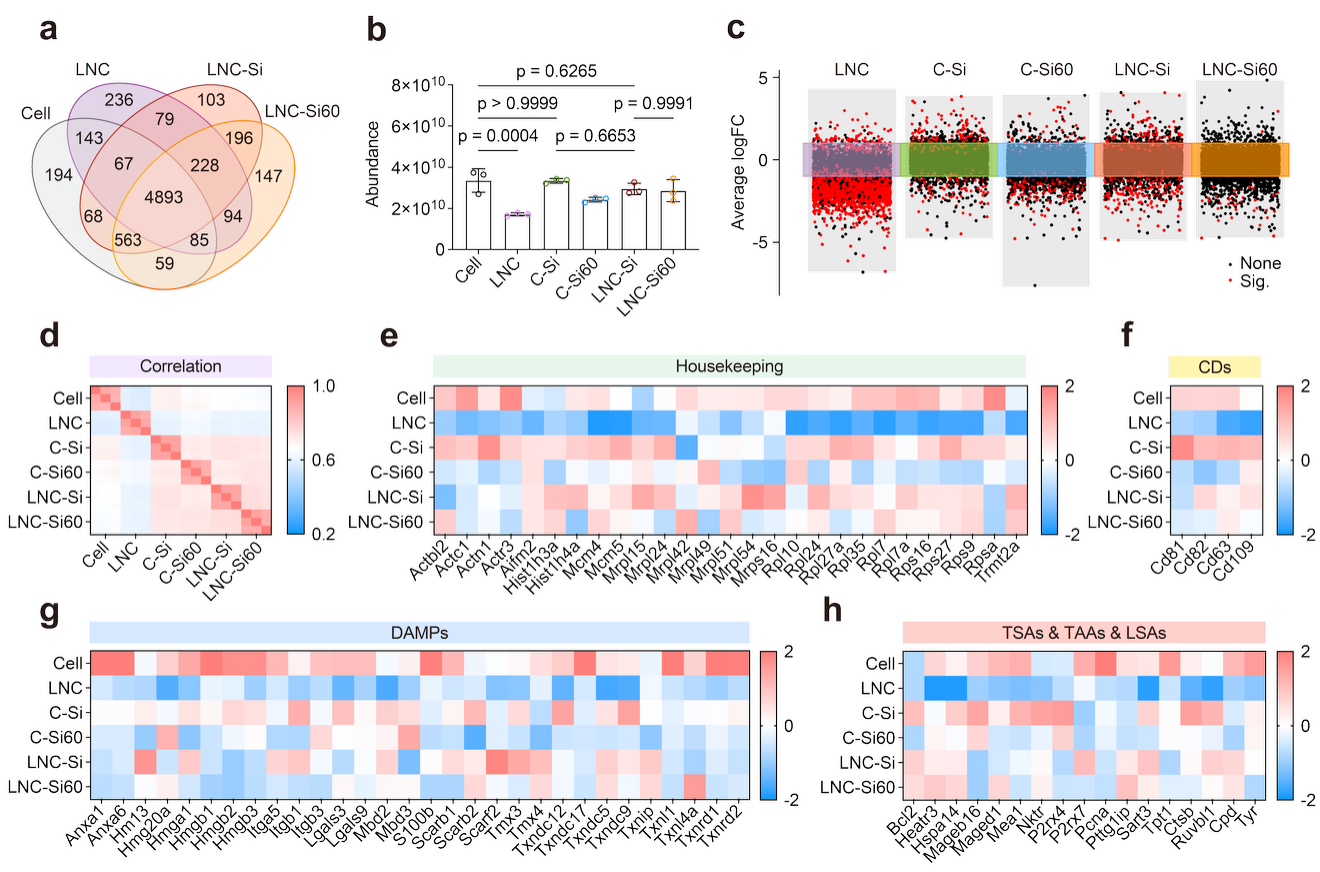
Figure 3. Protein protection in biomimetic cell vaccines.
In exploring the broad-spectrum prophylactic efficacy of heterogeneous vaccines, the team observed that individualized vaccination extended median survival by 35.8%, while heterogeneous vaccination extended it by 47.2%. The heterogeneous tumor vaccine achieved a prophylactic effect comparable to that of the individualized vaccine, supporting its broad applicability against tumor heterogeneity. Thus, biomimetic cell vaccines enable not only personalized tumor prevention but also universal protection against aggressive heterogeneous tumor subtypes, holding promise for preventing tumor recurrence in patients and advancing general tumor prevention in healthy populations.
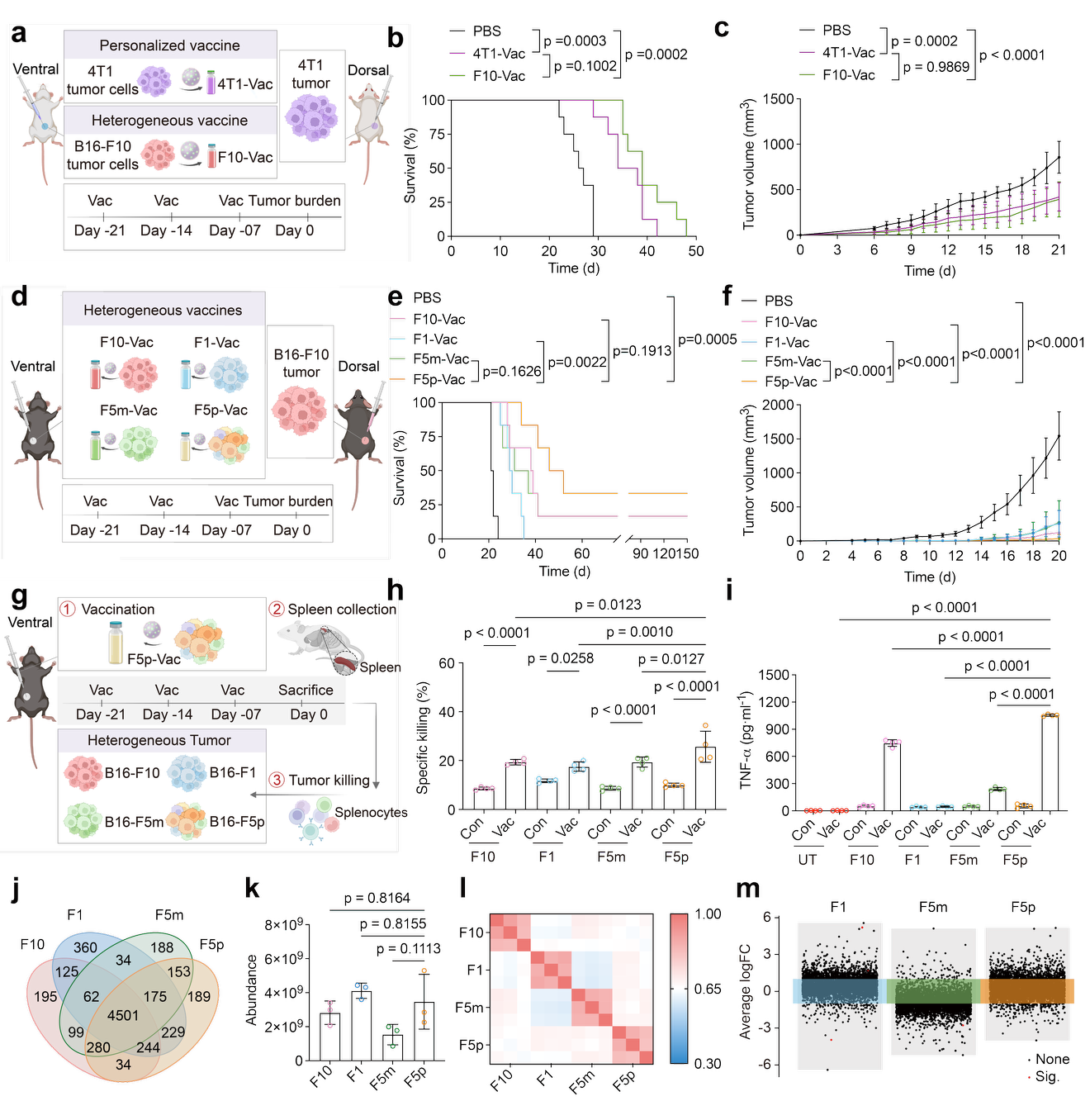
Figure 4. Biomimetic cell vaccines for universal immunotherapy in vivo.
Core highlights of this study:
(1) High-fidelity antigen preservation addressing tumor heterogeneity: Approximately 90% of cellular proteins are retained after cryopreservation, significantly outperforming conventional methods (~50% retention), thereby preserving the complex whole-tumor cell antigen repertoire.
(2) Construction of a multivalent universal antigen system with validated broad-spectrum prophylactic efficacy against heterogeneous tumors: Using heterogeneous tumor cell lines evolved under in vivo microenvironmental selective pressure, a "universal vaccine" covering a broad antigen spectrum was developed to effectively address inter-patient heterogeneity. The universal heterogeneous vaccine extended median survival by 2.28-fold and achieved complete tumor prevention in some individuals.
(3) Mechanically regulated immune response with significantly enhanced phagocytosis and antigen presentation by antigen-presenting cells: Mechanical reinforcement increased the phagocytic efficiency of antigen-presenting cells by 7.21-fold, accelerating immune processing of complex antigens. Further studies revealed that bone marrow-derived dendritic cells (BMDCs) can sense changes in cellular mechanical properties and transduce these cues into immune signals, thereby regulating the phagocytosis and presentation of heterogeneous antigens. This "mechano-immunological" coupling mechanism provides a new theoretical foundation for precisely modulating immune responses to complex antigens.
(4) Rapid preparation and translational potential: The vaccine can be prepared within six hours, greatly enhancing flexibility for clinical application.
This work was jointly supported by the Guangdong Basic and Applied Basic Research Foundation, the National Natural Science Foundation of China, the Pearl River Talent Program of Guangdong Province, the Program for Guangdong Introducing Innovative and Entrepreneurial Teams, the Natural Science Foundation of Guangdong Province, the Science and Technology Project of Guangzhou, the Fundamental Research Funds for the Central Universities of China, among other projects.