Professor Wei Zhu's team Publishes in Small: Programmable “Nano-Armored” Probiotics Enable Inflammation-Responsive Release and Precise Remodeling of the Gut Microecology
Recently, the team led by Professor Wei Zhu from the School of Biology and Biological Engineering, South China University of Technology, published a research article in the internationally renowned journal Small (IF = 12.1), entitled Programmable Biohybrid Probiotics with Long-TermStorage Stability for Enhanced Intestinal MicrobiotaRegulation and Ulcerative Colitis Treatment. The co-first authors are Jiani Jiang, a PhD student at South China University of Technology, and Jiangyan Dong, a master’s student at South China University of Technology. The corresponding authors are Professor Wei Zhu, Researcher Kaisheng Liu from Shenzhen People’s Hospital, and Associate Professor Quhuan Li.
Article link: https://onlinelibrary.wiley.com/doi/10.1002/smll.202512736
In the treatment of ulcerative colitis (UC), conventional drugs have long been limited by high recurrence rates, significant side effects, and insufficient local therapeutic efficacy. Although live probiotic therapy is regarded as a next-generation precision intervention strategy, its clinical application still faces three major bottlenecks: limited storage stability, monofunctional therapeutic effect, and insufficient drug exposure at inflamed lesions. How to enable probiotics to “arrive alive, release precisely, and function persistently” has become a key scientific challenge that urgently needs to be addressed in this field.
In the treatment of ulcerative colitis (UC), conventional drugs have long been constrained by high recurrence rates, significant side effects, and limited local therapeutic efficacy. Although live probiotic therapy is considered a next-generation precision intervention strategy, its clinical translation remains hindered by three major bottlenecks: limited storage stability, single-function therapeutic effects, and inadequate drug exposure at inflamed lesions. Enabling probiotics to “arrive alive, release precisely, and function persistently” has thus emerged as a critical scientific challenge that demands urgent attention.
Exogenous regulation of living organisms using functional components to enhance their structure and function has emerged as a promising strategy for biological engineering. Professor Wei Zhu’s group previously conducted extensive research based on this strategy (Adv. Mater. 2025, 37, 2417050; Adv. Mater. 2024, 2407831; PNAS 2024, 121, e2408273121; PNAS 2024, 121, e2322418121; Angew. Chem. Int. Ed. 2024, 63, e20240611). Recently, Professor Wei Zhu’s team, in collaboration with Researcher Kaisheng Liu, innovatively constructed a programmable biohybrid probiotic platform, Bif@FCSM(A). Using Bifidobacterium as the core carrier, this platform, for the first time, integrates structural reinforcement, functional customization, and responsive release through a multilayer modular assembly strategy, endowing probiotics with enhanced environmental adaptability and precision therapeutic capability. This platform effectively overcomes key bottlenecks in the clinical application of anaerobic probiotics (such as Bifidobacterium longum), significantly improving storage stability, oral delivery efficiency, and therapeutic versatility, and provides a new technical route for the clinical translation of live probiotic therapies.
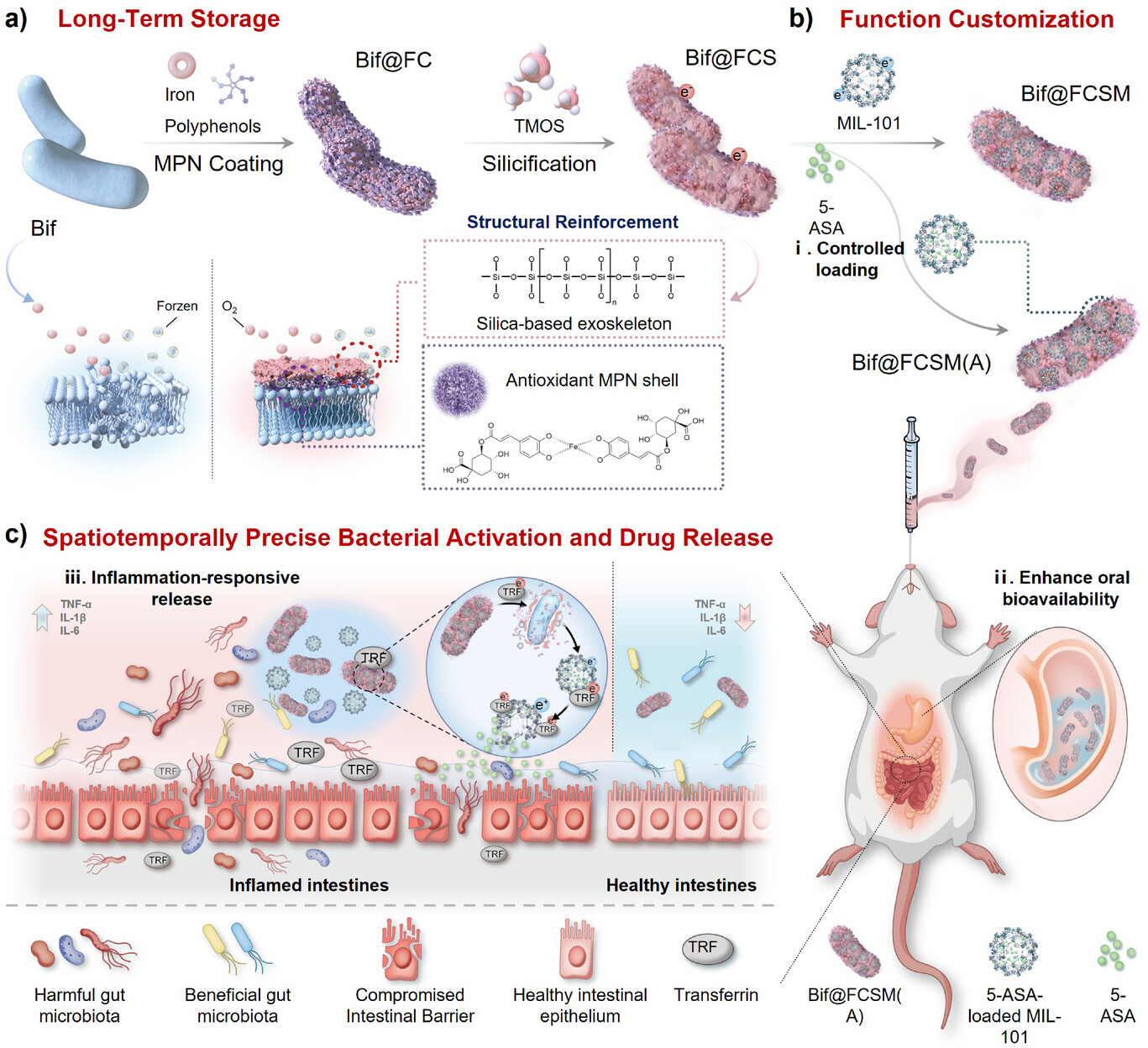
Figure 1. Schematic illustration of the programmable biohybrid probiotic platform for alleviating ulcerative colitis. a) Long-term storage performance; b) Functional customizability; c) Spatiotemporally precise bacterial activation and drug release.
The highlights of this study include:
(1)Structural reinforcement: multilayer “nano-armor” markedly improves stability
A multilayer modular encapsulation system consisting of an MPN oxygen-shielding layer + SiO₂ structural reinforcement layer was constructed to effectively resist oxidative damage and gastric acid attack. Under aerobic storage conditions, probiotic viability was increased by 41-fold, and survival in gastric fluid was enhanced by 871-fold, significantly overcoming the stability bottleneck of conventional live bacterial delivery.
(2)Functional customization: modular drug loading enables precise probiotic functionalization
The high specific surface area and large pore volume of MIL-101-Fe were utilized to achieve highly efficient loading of therapeutic agents, endowing the bacteria with programmable therapeutic functionality. This design upgrades the bacteria from a single microbiota-regulating agent to an intelligent therapeutic carrier with dual functions of live bacterial therapy and drug delivery.
(3)Responsive release: lesion microenvironment-triggered precise activation therapy
Highly expressed transferrin in inflamed tissues competitively chelates Fe³⁺, triggering the disassembly of the outer structure and enabling precise release and synchronous activation of both drugs and probiotics at the lesion site, thereby upgrading the treatment mode from passive delivery to lesion-driven intelligent responsiveness, with significantly improved local therapeutic efficiency.
This work is the first to organically integrate long-term storage stability, inflammation-responsive release, and precise microbiota regulation into a single probiotic platform, providing a new design paradigm for next-generation intelligent live bacterial therapeutic systems.
This work was financially supported by Shenzhen Science and Technology Program, Program of Marine Economy Development Special Fund under Department of Natural Resources of Guangdong Province, the National Natural Science Foundation of China, and Five Three Program of Shenzhen People's Hospital, the National Natural Science Foundation of China, Guangdong S&T Program, Guangzhou Science and Technology Plan Project, and the Fundamental Research Funds for the Central Universities of China.