Professor Wei Zhu Published Paper in Materials Today: Squaric Ester-based Haptenized Whole Tumor Cell Vaccines for Enhanced Cancer Immunotherapy
Recently, Professor Wei Zhu from the School of Biology and Biological Engineering at South China University of Technology, in collaboration with Professor Yanli Zhao from Nanyang Technological University, Singapore, published a research paper entitled Haptenized whole tumor cell vaccines for cancer immunotherapy online in Materials Today (IF=21.1), a world-renowned academic journal. Dr. Yun Chen, a postdoctoral fellow at the School of Biology and Biological Engineering of South China University of Technology, is the first author of the paper, and Professor Wei Zhu and Professor Yanli Zhao are the co-corresponding authors.
Link to the paper:https://www.sciencedirect.com/science/article/pii/S136970212600074X
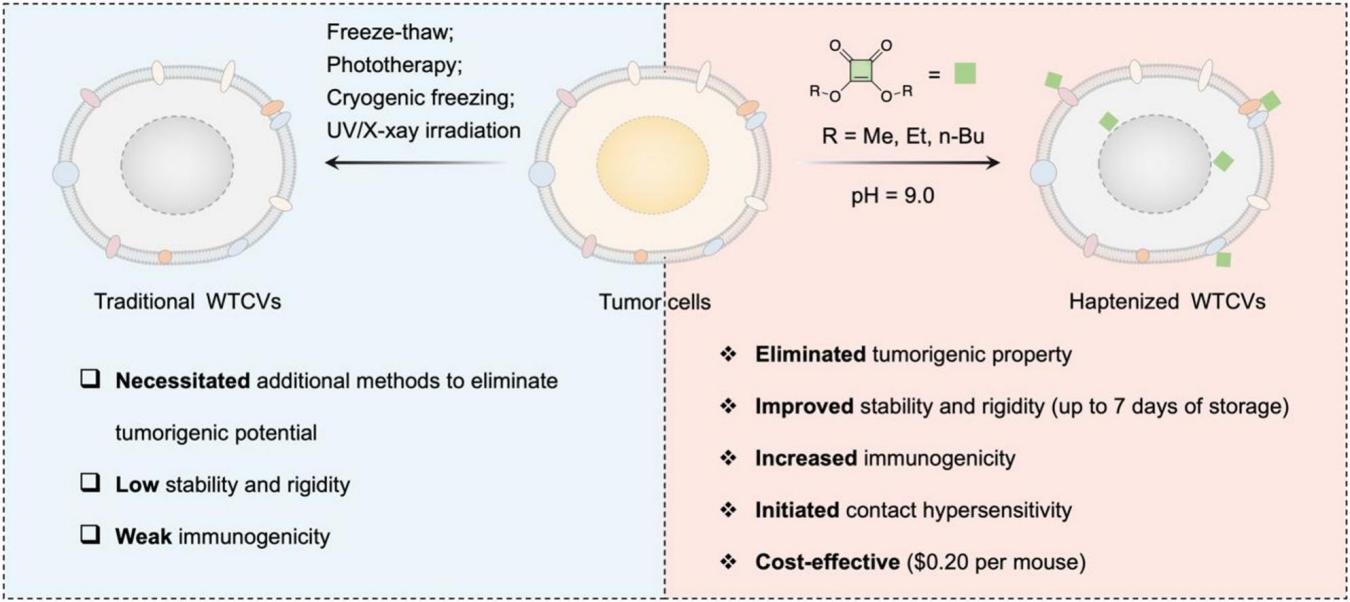
Figure 1. Strategy and advantages of fabricating squaric ester-based haptenized whole tumor cell vaccines (SE-Vs).
Tumor vaccines have shown tremendous potential in revolutionizing oncological therapy by harnessing the body's immune system. Among them, Whole Tumor Cell Vaccines (WTCVs) are regarded as highly promising candidates, as they contain a diverse and comprehensive array of tumor antigens and can activate innate immunity, subsequently triggering antigen-specific adaptive immune responses. However, in pivotal clinical trials, these vaccines have only achieved limited objective response rates and have exerted a minimal effect on improving patient survival. This is mainly attributed to the insufficient tumor antigen expression and blocked antigen presentation caused by the immune evasion mechanisms of tumor cells. In addition, although various strategies have been developed to enhance the immunogenicity of WTCVs, their preparation process remains complex and time-consuming (usually more than 24 hours), and requires additional methods such as repeated freeze-thaw cycles, ultraviolet (UV) or X-ray irradiation to eliminate their tumorigenic potential. Therefore, there is an urgent need to develop a simple, safe and innovative vaccine preparation method to enhance antigen recognition and address the deficiencies in antigen presentation.
A hapten is a class of small molecules with a molecular weight typically less than 1000 Da, which cannot induce an immune response independently on its own. However, when conjugated to a large macromolecular carrier protein, haptens can significantly enhance immunogenicity, providing an important approach to improve the efficacy of WTCVs. In this study, the research team innovatively utilized squaric ester compounds to haptenize whole tumor cells, and developed a novel type of WTCVs termed SE-Vs with an extremely simple preparation process and ultra-low cost. The vaccine can be fully prepared within only 2 hours, with the chemical raw material cost of a single mouse dose at approximately $0.20. Meanwhile, it can completely eliminate the tumorigenic potential of tumor cells, induce immunogenic cell death (ICD) in tumor cells, and stably retain its immune activity after 7 days of lyophilized storage, greatly breaking through the limitations of traditional vaccines in terms of preparation cycle, storage cost and shelf life.
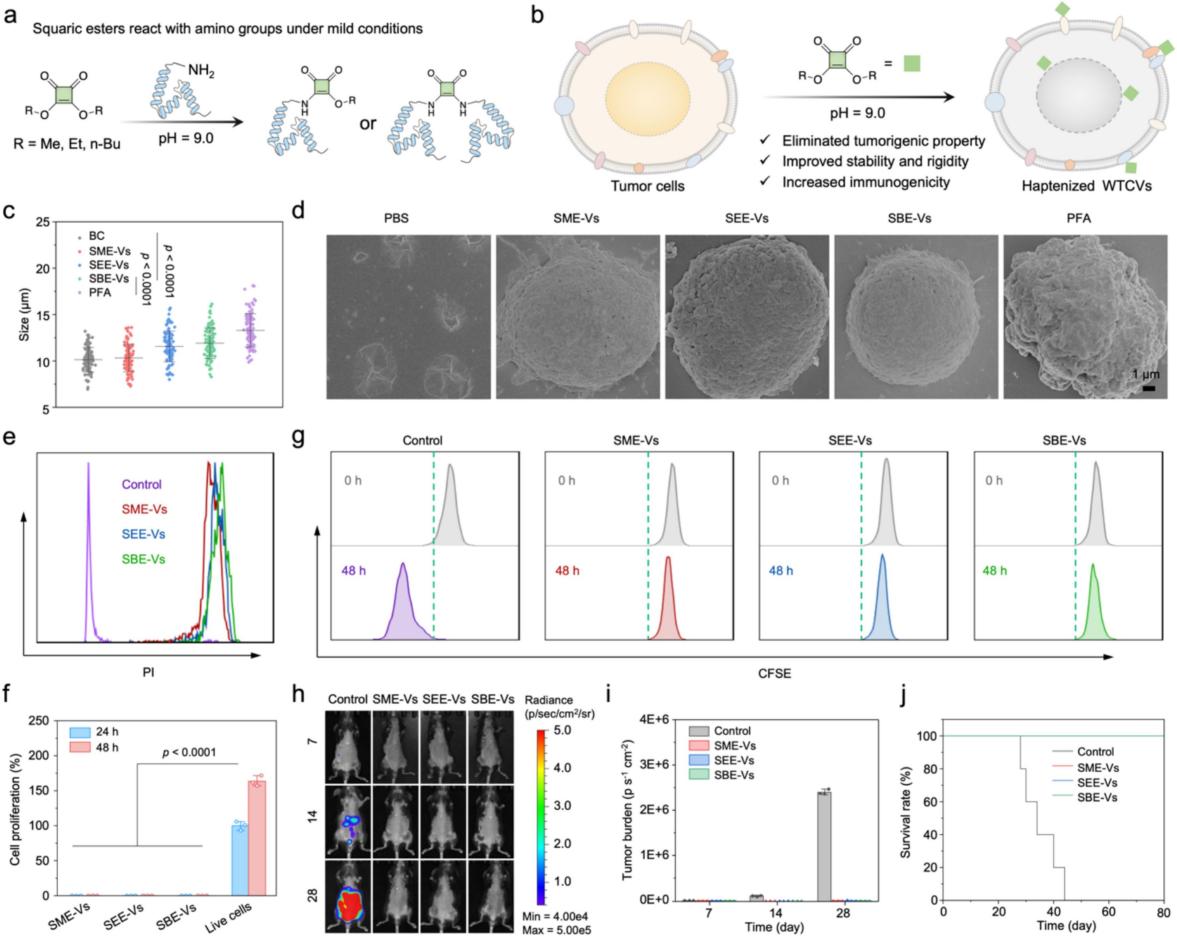
Figure 2. Preparation and characterization of squaric ester-haptenized whole tumor cell vaccines (SE-Vs). a) Molecular mechanism of the mono- or dual-ligation reaction between squaric esters and amino groups of proteins on the cell surface or within cells; b) Core workflow for the preparation of corresponding vaccines using three squaric ester derivatives, namely Squaric Acid Dimethyl Ester (SADME), Squaric Acid Diethyl Ester (SADEE), and Squaric Acid Dibutyl Ester (SADBE), which are SADME-haptenized Whole Tumor Cell Vaccines (SME-Vs), SADEE-haptenized Whole Tumor Cell Vaccines (SEE-Vs), and SADBE-haptenized Whole Tumor Cell Vaccines (SBE-Vs), respectively; c-g) Relevant characterization results of the cell morphology, structural integrity, inactivation effect and tumorigenicity elimination of SE-Vs, confirming that squaric ester modification can effectively preserve the intact cell structure and completely eliminate the proliferation capacity of tumor cells.
The core highlights of this study include:(1) Minimalist preparation and ultra-high stability: The whole preparation process of the vaccine can be completed within 2 hours without additional inactivation steps; the dual-ligation reaction of squaric esters endows the vaccine with ultra-high structural stability, which can maintain the integrity of cell structure in pure water for up to 100 days, and retain full immune activity after 7 days of lyophilized storage, with a validity period far exceeding that of similar vaccine products reported previously.(2) Efficient induction of immunogenic cell death: Squaric ester modification can significantly increase the extracellular exposure level of Calreticulin (CRT), a key marker of immunogenic cell death, by up to 3.1 times that of the control group. Meanwhile, it completely retains tumor-specific antigens and immune regulation-related molecules, effectively activating the body's innate immune response.(3) Potent promotion of antigen cross-presentation: Haptenization modification can induce Contact Hypersensitivity (CHS), construct a multicellular immune niche, and significantly promote the maturation of dendritic cells (DCs) and M1-type polarization of macrophages. Among them, the SBE-Vs group with the best performance showed significantly higher antigen cross-presentation efficiency than the control group, laying a core foundation for the robust activation of antigen-specific CD8+ T cell responses.(4) Broad-spectrum universality and high biosafety: This strategy has been fully validated in a variety of tumor cell lines including melanoma, breast cancer, colon cancer and lung cancer, with good broad-spectrum applicability; in vivo experiments confirmed that the vaccine has no obvious systemic toxicity, and the body weight of mice remained stable during the treatment cycle, with excellent biosafety for clinical translation.
After a systematic comparative study of vaccines modified with the three squaric ester derivatives, the research team found that SBE-Vs, the vaccine modified with squaric acid dibutyl ester, exhibited the optimal anti-tumor efficacy. In the prophylactic immunization experiment, SBE-Vs achieved a tumor onset inhibition rate of up to 87.0% in healthy mice, providing robust protection against tumor challenge for the body. In therapeutic application scenarios, SBE-Vs effectively induced tumor regression, with 20% of the treated mice achieving long-term tumor-free survival for at least 70 days. When combined with Doxorubicin (DOX), a first-line clinical chemotherapeutic drug, the tumor growth inhibition rate reached 73.1%, demonstrating excellent combination therapeutic potential.
This research achievement represents a key breakthrough in WTCV technology. It not only greatly simplifies the vaccine preparation process and reduces the cost of clinical application, but also mechanistically solves the core pain points of traditional WTCVs, including insufficient immunogenicity and low antigen presentation efficiency. It broadens the application boundary of hapten-based immunotherapy, provides a brand-new research perspective and candidate solution for clinical tumor immunotherapy, and possesses extremely high clinical translation value and application prospects.
This work was jointly supported by the Guangdong Basic and Applied Basic Research Foundation, the National Natural Science Foundation of China, the Pearl River Talent Program of Guangdong Province, the Program for Guangdong Introducing Innovative and Entrepreneurial Teams, the Natural Science Foundation of Guangdong Province, the Science and Technology Project of Guangzhou, the Fundamental Research Funds for the Central Universities of China, a funding from the College of Science at Nanyang Technological University, and the National Research Foundation Singapore, among other projects.