Professor Wei Zhu Published a Perspective Paper in ACS Nano: Biosilicification Across Biological Hierarchies
Recently, Professor Wei Zhu from the School of Biology and Biological Engineering at South China University of Technology published a perspective paper entitled Biosilicification across biological hierarchies online in ACS Nano (IF=16.5), a top-tier journal in nanoscience. The paper systematically elaborates on the research progress, core systems, and translational prospects of biosilicification technology across all hierarchical scales from biomolecules to macroscopic organisms, opening up a new direction for interdisciplinary fields including bioinorganic engineering, biological resource preservation, and cancer immunotherapy. The co-first authors of the paper are Muyuyang Lin, a master’s student, Sishi Guo, a doctoral student, and Dr. Liang Zhou, a postdoctoral researcher, all from the School of Biology and Biological Engineering at South China University of Technology. Professor Wei Zhu and Academician C. Jeffrey Brinker from the University of New Mexico, USA, are the co-corresponding authors.
Link to the paper: https://pubs.acs.org/doi/10.1021/acsnano.5c22206
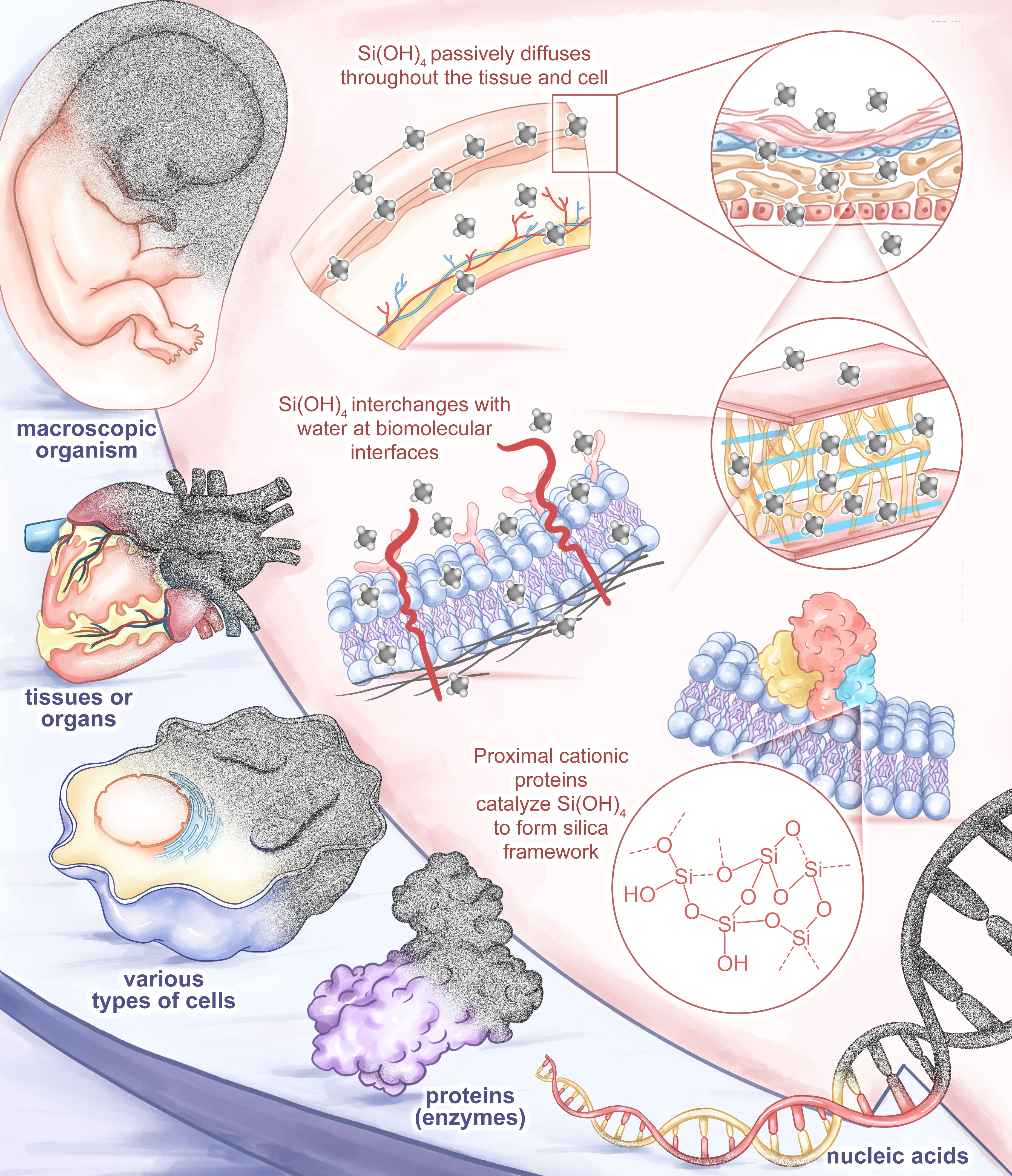
Figure 1.Schematic illustration of biosilicification technology for stabilizing or functionally evolving biological specimens across different hierarchical scales.
The long-term stable preservation and functional enhancement of biological samples and bioactive substances have long been core challenges in the fields of life sciences and biomedicine. Traditional methods, such as ultra-low temperature cryopreservation and chemical fixation, have many bottlenecks, including high cost, heavy reliance on cold chains, and irreversible damage to biological structures and genetic information. In contrast, siliceous organisms such as diatoms in the ocean have evolved sophisticated regulatory pathways for biosilicification over hundreds of millions of years, enabling them to construct highly ordered silica bioskeletons with exceptional mechanical properties under mild physiological conditions. This provides a natural biomimetic blueprint for the development of artificial biosilicification technology. In recent years, with the in-depth interdisciplinary integration of synthetic biology, materials science, and bioinorganic chemistry, biosilicification technology has achieved leapfrog development and become a core direction to break through the bottlenecks of traditional biological preservation and functional enhancement technologies.
What is “Biosilicification”?
Biosilicification refers to the biologically regulated process of uptake, transport, and deposition of silicon, which can in situ form silicon-based biomineral structures inside or on the surface of living systems through protein-catalyzed sol-gel reactions, representing a type of organic-inorganic hybrid interface engineering. Compared with the traditional material application mode of “preparation in vitro-delivery in vivo”, biosilicification has irreplaceable core advantages: mild reaction conditions, with the whole process carried out in an aqueous environment at ambient temperature and pressure without severe physical and chemical stimulation; strong cross-scale universality, covering full hierarchical samples from biomacromolecules such as nucleic acids and proteins to intact organs and multicellular organisms; self-limiting deposition, enabling precise regulation of the thickness and porosity of the silica layer to balance the protective effect and material exchange; excellent biocompatibility, with silicified products capable of controllable degradation in vivo and safe clearance via the kidneys without long-term accumulation toxicity.
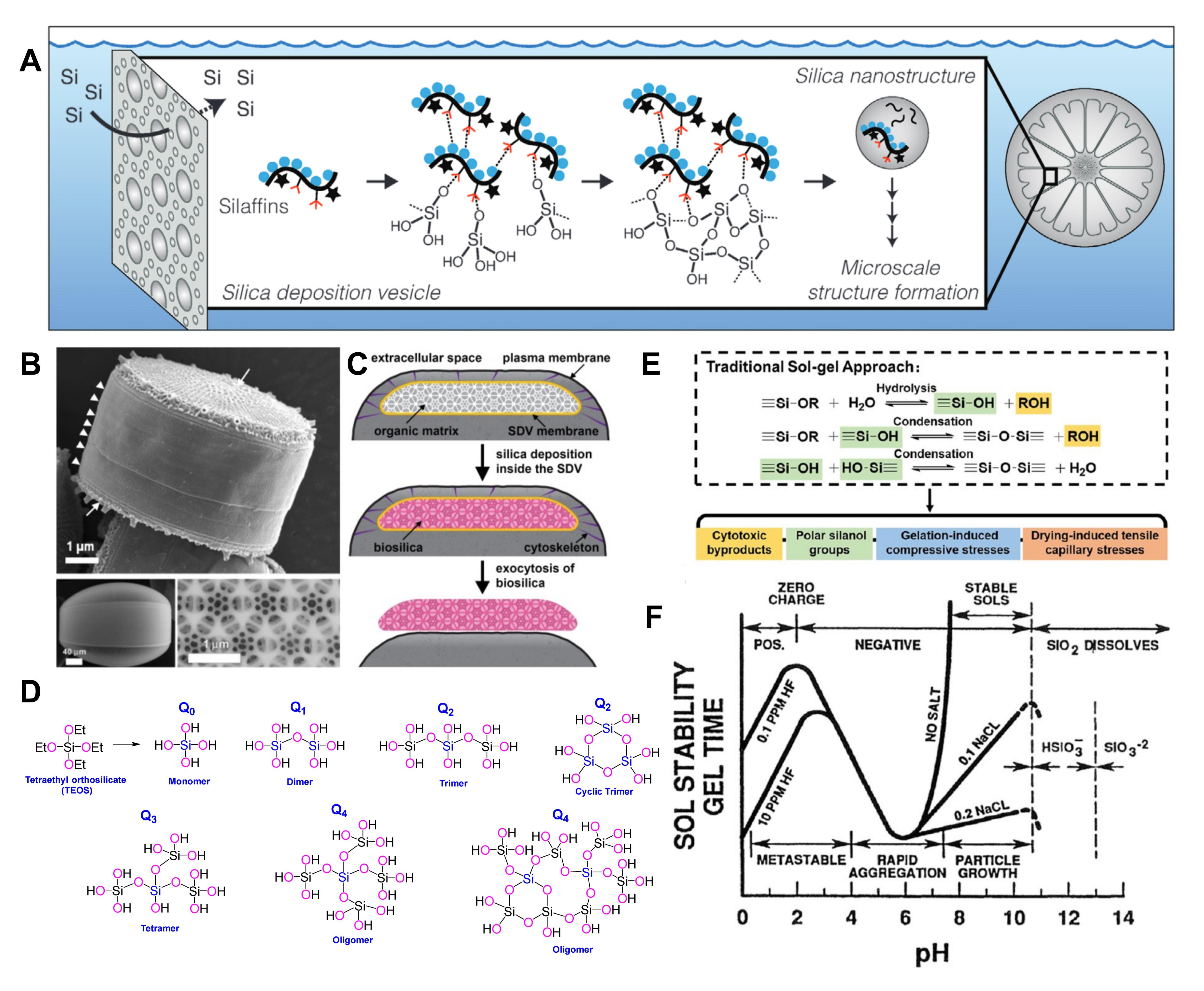
Figure 2. Natural biosilicification and artificially customized biomimetic silicification technology.
From Nucleic Acid Molecules to Whole Organisms: Four Core Application Hierarchies
Biosilicification at the Nucleic Acid Scale
As the core carrier of genetic information, the stable preservation of nucleic acids is critical to maintaining the continuity and diversity of biological heredity. However, native Deoxyribonucleic Acid (DNA) has a fragile structure and is highly susceptible to degradation by factors such as temperature, enzymatic hydrolysis, and oxidation. Biosilicification at the nucleic acid scale focuses on achieving in situ encapsulation and high-fidelity protection of genomic DNA through technologies such as cryosilicification and deep silicification. Via optimized delivery strategies for silica precursors, orthosilicic acid molecules can efficiently penetrate into cells and nuclei, forming a uniform silica network catalyzed by proteins to achieve 360° encapsulation and protection of the whole genome. For example, cryosilicification technology can extend the half-life of genomic DNA at ambient temperature to 1,208 years, 167 times that of unprotected samples, while maintaining an extremely low gene mutation rate even lower than the error rate of Taq polymerase in Polymerase Chain Reaction (PCR) amplification. Deep silicification technology realizes cross-scale genome preservation from microorganisms to multicellular organisms, with the recovery rate of target gene fragments still exceeding 50% after 3 weeks of accelerated aging at 70 ℃, providing a disruptive solution for the construction of biological germplasm resource banks, forensic sample preservation, and paleogenomic research.
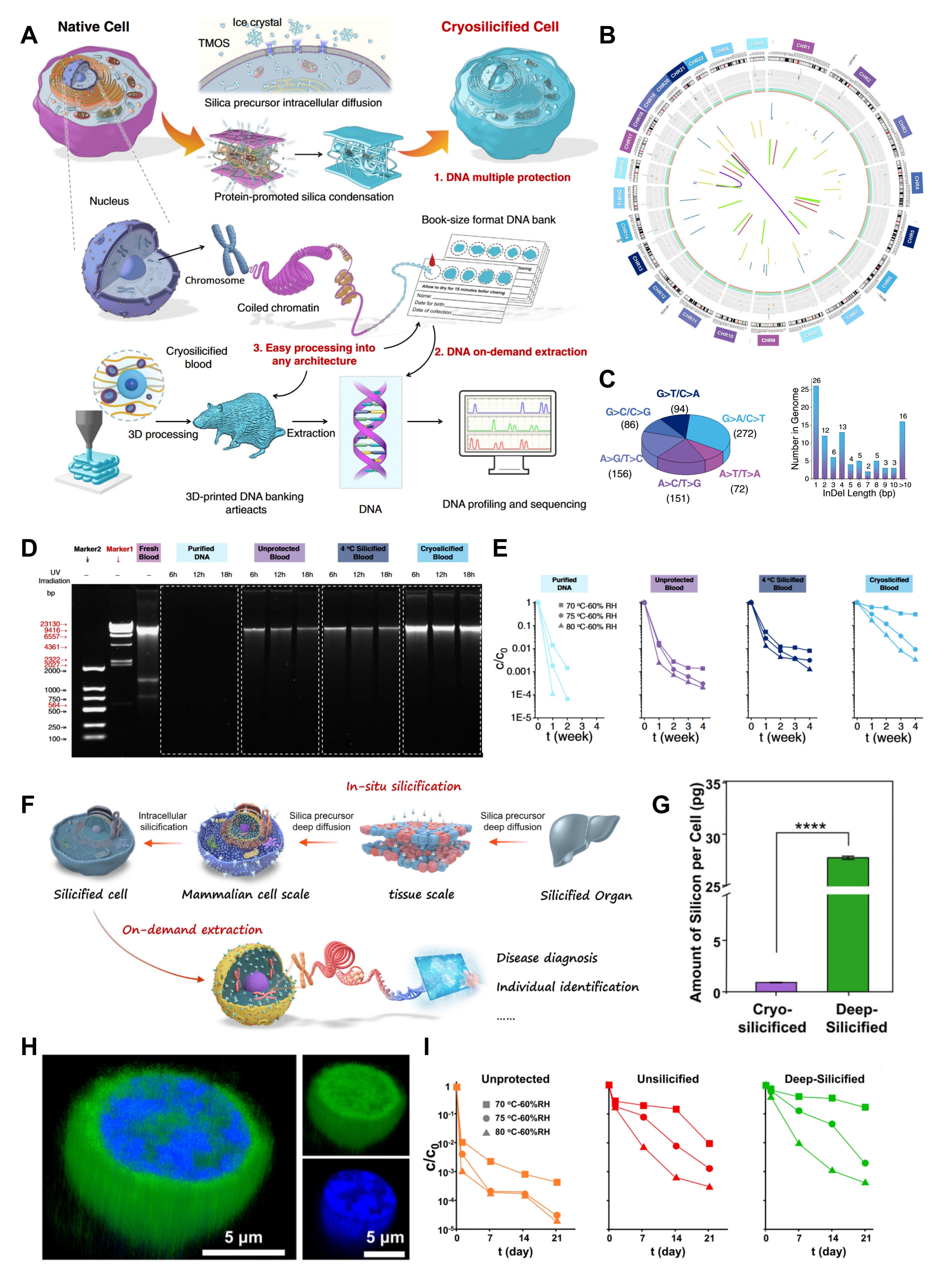
Figure 3. Biosilicification at the nucleic acid scale.
Biosilicification at the Protein (Enzyme) Scale
Proteins and enzymes are the core executors of life activities, but their native structures are extremely unstable and prone to denaturation and inactivation under environmental stress, which has long made them highly dependent on cold chain systems for preservation and transportation. Biosilicification at the protein scale achieves full-dimensional protection and functional enhancement from extracellular single enzyme molecules to intracellular proteomes. At the single-enzyme level, the innovatively developed “silicazyme” technology, through precise modification of multiple silica precursors on single enzyme molecules, breaks the zero-sum dilemma of “trade-off between activity and stability” in traditional enzyme immobilization. It not only increases the catalytic activity of industrial enzymes such as lipase and glucose oxidase by 1.26-1.95 times, but also greatly enhances their resistance to high temperature, organic solvents, and extreme pH. For intracellular proteins, self-limiting silicification technology can form a nanoscale semipermeable silica shell inside cells, with more than 80% of the activity of various key intracellular enzymes retained after 1 year of storage at room temperature, compared with less than 3% retention by traditional fixation methods, completely eliminating the dependence of biological enzyme preparations on cold chainsystems.
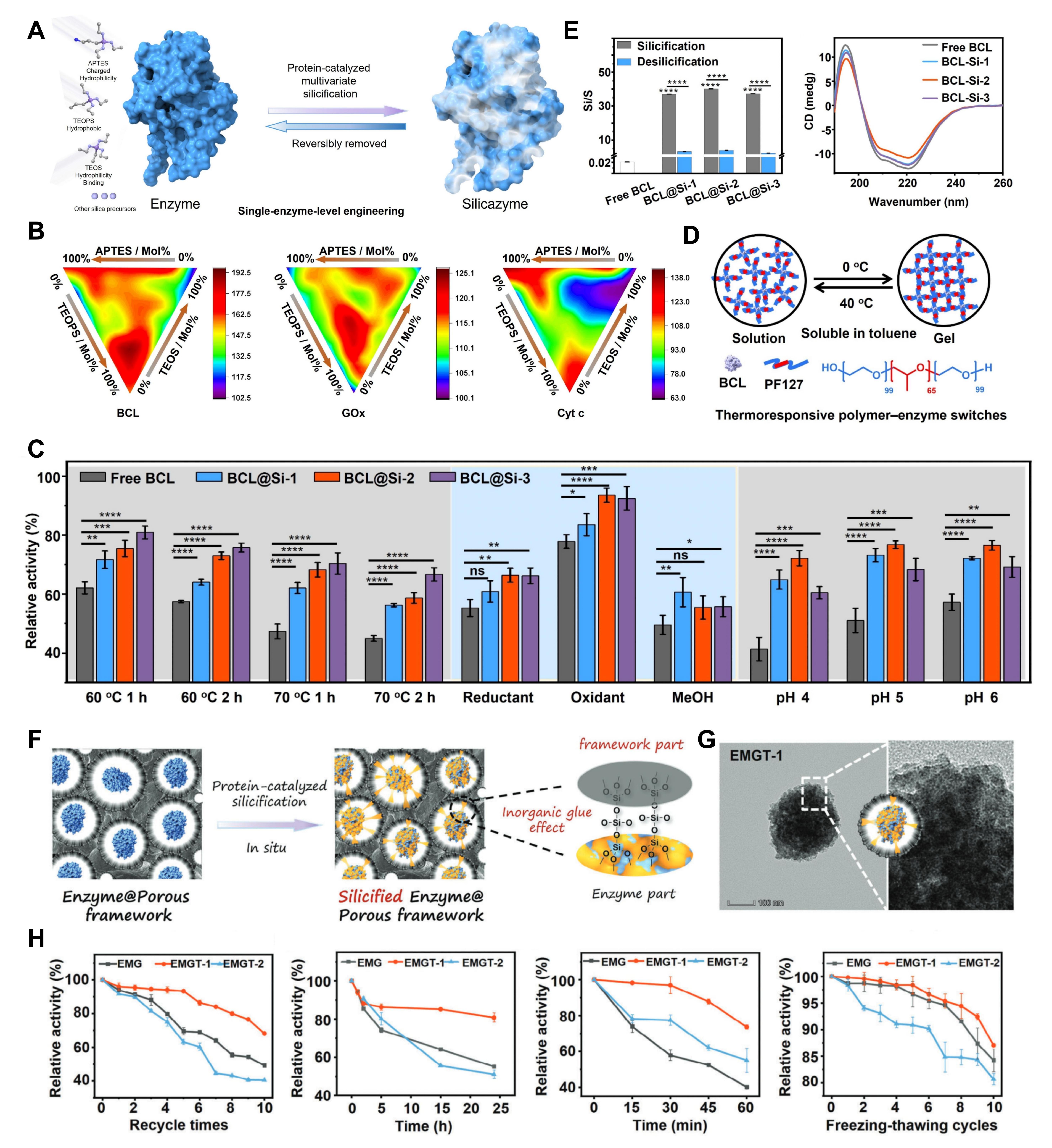
Figure 4. Biosilicification at the protein scale.
Biosilicification at the Cellular Scale
Cells are the basic structural and functional units of life, as well as the core carriers for cell therapy, regenerative medicine, and vaccine development, making biosilicification at the cellular scale the core application scenario of this technology. Through controllable silicification on the cell membrane surface or inside cells, it can not only build an “inorganic armor” for cells to enhance environmental tolerance, but also precisely regulate the physiological functions and fate of cells to adapt to diverse biomedical applications. For example, in the field of tumor immunotherapy, whole-cell tumor vaccines developed based on silicification technology can stably preserve whole tumor antigens for a long time under dry conditions at room temperature. After functional modification, the vaccine achieved a 100% cure rate in a mouse ovarian cancer model, and a single prophylactic immunization provided complete protection against tumor challenge for the body. In the field of transfusion medicine, biomimetic silicification on the surface of red blood cells can effectively shield blood group antigens, constructing a new type of red blood cell product with universal transfusion potential, while fully retaining its oxygen-carrying capacity and in vivo circulation characteristics. In addition, biosilicification at the cellular scale can also achieve targeted apoptosis induction of tumor cells and directional regulation of osteogenic differentiation of mesenchymal stem cells (MSCs), providing a brand-new strategy for regenerative medicine and precision tumor therapy.

Figure 5. Biosilicification at the cellular scale.
Biosilicification at the Macroscopic Organism/Tissue Scale
High-fidelity long-term preservation of soft tissues, intact organs, and whole biological specimens has long been a major challenge in clinical pathology and biodiversity conservation. Traditional fixation methods are prone to cause sample structural deformation, loss of biological information, and imaging artifacts. Biosilicification technology at the macroscopic scale breaks through the tissue penetration bottleneck of silica precursors, achieving full-structure high-fidelity preservation from isolated soft tissues and intact organs to multicellular model organisms. Silica bioreplication technology can form a uniform nanoscale silica layer on intact biological samples such as chicken embryos, preserving full hierarchical biological features from subcellular ultrastructure to the overall organ morphology with high fidelity. Deep silicification technology further realizes the simultaneous preservation of structural and genomic information of macroscopic organisms such as intact mouse organs and earthworms, with an estimated storage life of nearly a thousand years for samples at ambient temperature, and 96.36% of the complete genomic information retained after simulated storage for nearly a century, providing a brand-new technical solution for the construction of clinical biological sample banks, biological germplasm resource banks, and museum specimen preservation.
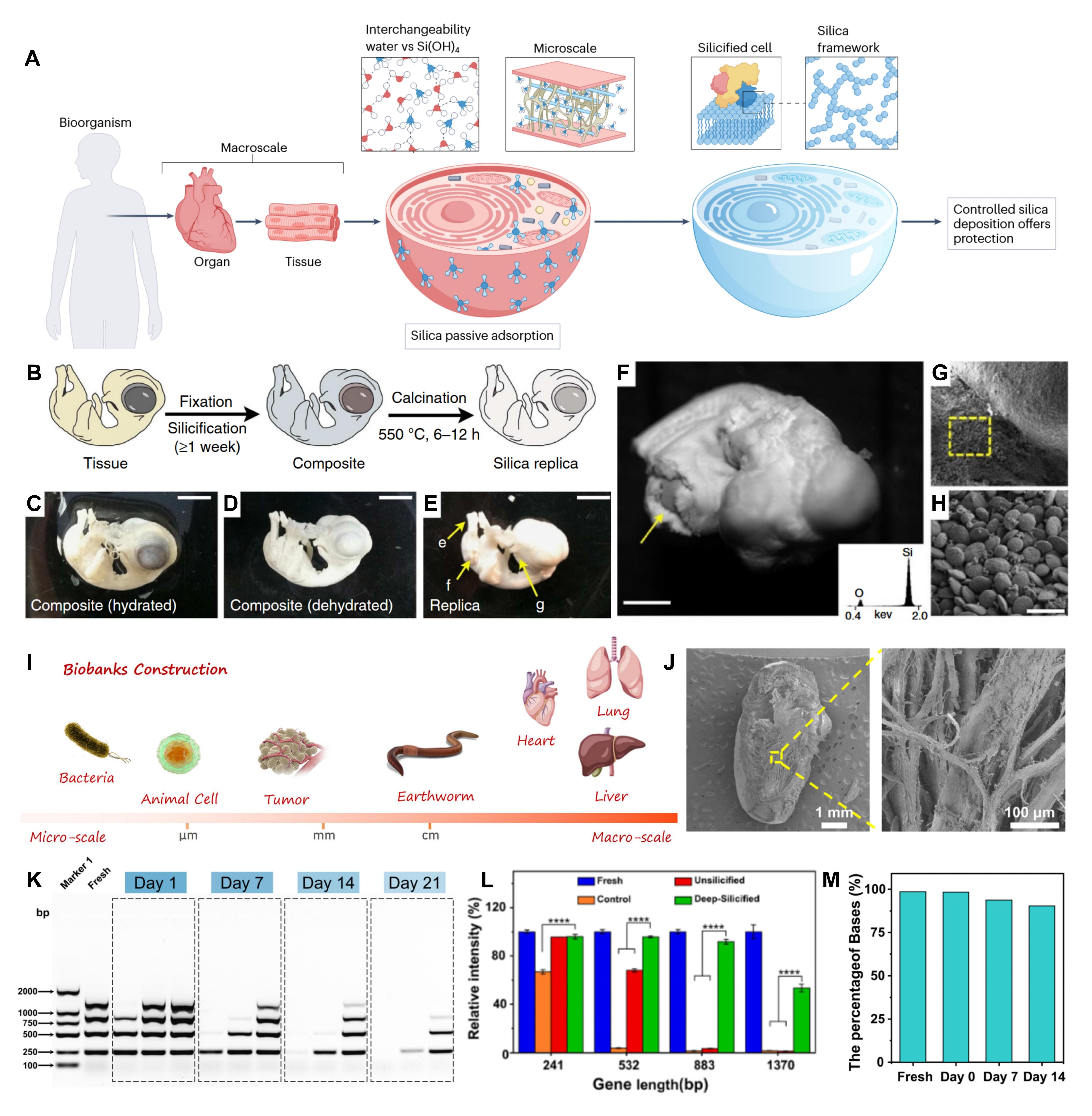
Figure 6. Biosilicification at the macroscopic scale of organs, tissues, and whole organisms
Promising Prospects Alongside Challenges
Although biosilicification technology across biological hierarchies has shown extremely broad application prospects in biological preservation, biomedicine, regenerative medicine and other fields, it still faces many core challenges in the process of clinical translation and large-scale application: the penetration efficiency and deposition uniformity of silica precursors in complex biological tissues still need to be further optimized; the in vivo degradation kinetics and long-term metabolic fate of silicified products at different hierarchies need to be more systematically elucidated; the spatiotemporal precise control of the silicification reaction and its matching with the physiological window in vivo are still not perfect; the standardized preparation process and quality control system for clinical applications have not yet been established.
In response to these challenges, the paper also proposes four key development directions worthy of focus in the future: combining directed evolution in synthetic biology and Artificial Intelligence (AI) protein design technology to develop silicification regulatory elements with higher catalytic efficiency and stronger targeting, realizing programmable and precise regulation of the silicification process from the molecular scale to the tissue scale; expanding the application of biosilicification in more immune cell therapy platforms such as Natural Killer (NK) cells and macrophages to develop new universal cell therapy products; combining immune regulatory molecules such as immune checkpoint inhibitors and Stimulator of Interferon Genes (STING) agonists to construct synergistic anti-tumor immunotherapy strategies based on silicification technology; developing reversible and dynamically responsive biosilicification systems to achieve precise and reversible regulation of the functions of living systems, promoting a paradigm shift from “static structural preservation” to “dynamic functional regulation”.
“Biosilicification across biological hierarchies” is not a simple material synthesis technology, but an innovative achievement of the in-depth interdisciplinary integration of polymer chemistry, materials science, synthetic biology, and immunobiology. It breaks the traditional boundary between inorganic materials and living systems, injects new possibilities into the preservation of biological resources, the development of biomedicine, and the research of regenerative medicine, and promotes related fields to a new stage of precision, high efficiency, and universality. In the future, this technology will surely embark on a broad path with both scientific innovation value and industrial transformation potential through the collaborative development of multiple disciplines.
This work was financially supported by the National Natural Science Foundation of China (Grant Nos. 22372061, 22572061), Guangdong Science and Technology Program (Grant No. 2024B1111130002), Guangzhou Science and Technology Plan Project (Grant No. 2024A03J0163), and the Fundamental Research Funds for the Central Universities of China.