Professor Jianling Xie's Team Publishes a Research Article in Oncogene: Unveiling the Molecular Mechanism of Serine Protease-Driven Colorectal Cancer Progression, Providing a New Strategy for Targeted Therapy
Recently, a collaborative research team led by Professors Jianling Xie from the School of Biology and Biological Engineering, South China University of Technology (SCUT), and Kaikai Shen from Shanghai University of Traditional Chinese Medicine, published a research paper entitled PRSS22 inhibits HMOX1-mediated ferroptosis and induces osteopontin cleavage to promote M2 macrophage polarization and colitis-associated carcinogenesis in the authoritative oncology journal Oncogene. This study is the first to systematically elucidate the key driving role and molecular mechanism of the serine protease PRSS22 in the development and progression of inflammation-associated colorectal cancer (CRC), providing a novel theoretical basis and a potential target for the diagnosis and targeted therapy of CRC. The School of Biology and Biological Engineering, SCUT, is the primary corresponding institution of the paper. Professors Jianling Xie and Kaikai Shen serve as the corresponding authors. Zijian Kuang, a master's student from Professor Jianling Xie's research group, is the first author; and a PhD student from the same group, Wenken Liang, is the third author.

Article online link: https://www.nature.com/articles/s41388-026-03729-5#Sec24
CRC ranks among the most prevalent malignant tumours globally with high morbidity and mortality, causing nearly one million deaths annually. Inflammation-cancer transition is the core pathological process underlying the development of CRC. However, the therapeutic efficacy of current targeted therapies against this process remains limited, highlighting an urgent need to identify new key regulatory factors and their underlying mechanisms that aid disease progression. Members of the serine protease family play crucial roles in tumour initiation and progression, and their abnormal expression reshapes the cancer cell proteome to meet the demands of malignant proliferation. As a member of this protease family, PRSS22 has been implicated in various types of tumours, but its specific functions in CRC remained unclear prior to this study.
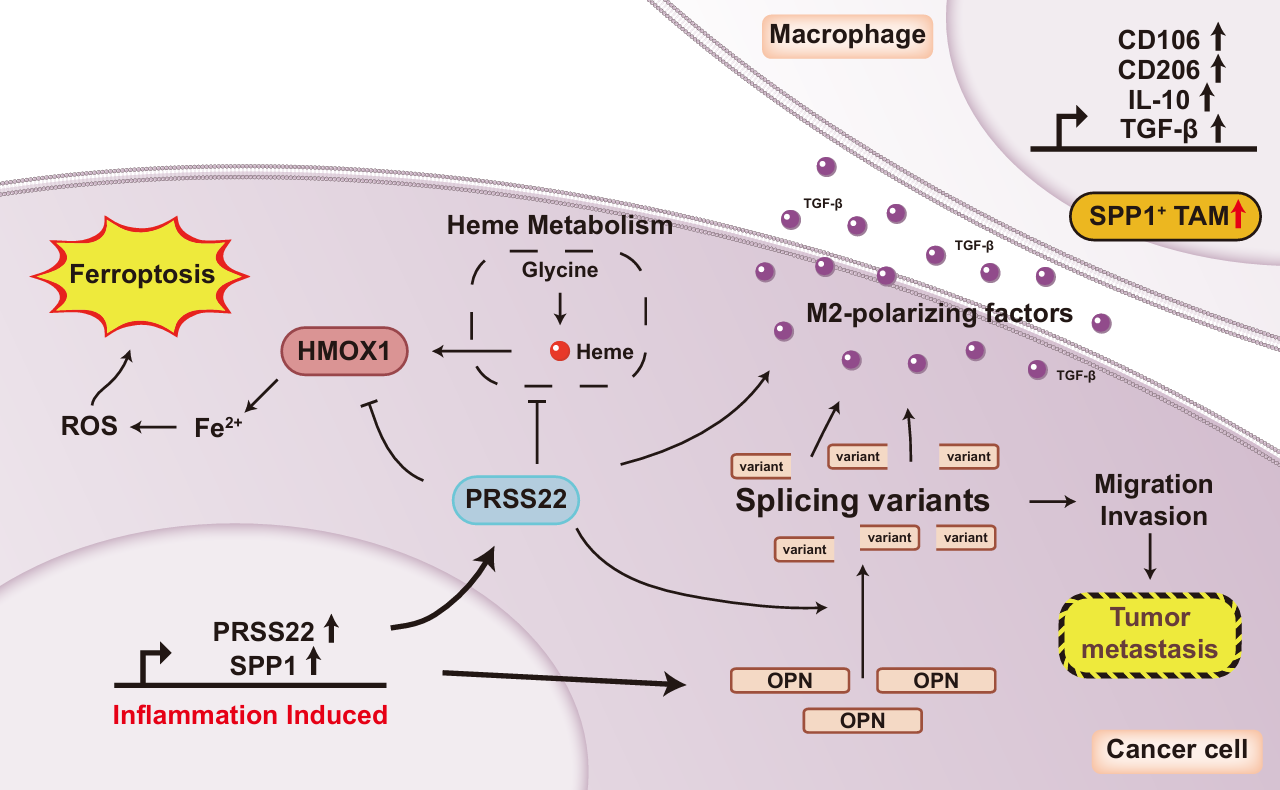
Figure 1: PRSS22 drives colitis-associated carcinogenesis through three pathways: inhibiting HMOX1-mediated ferroptosis, cleaving osteopontin (OPN) and promoting M2 macrophage polarization.
By integrating and analysing multi-omics data of colitis and CRC, the research team found that PRSS22 is significantly upregulated in the tissues of patients with inflammatory bowel disease and CRC. Moreover, its expression level is closely associated with tumour pathological progression, metastatic potential, and poor prognosis of patients. Receiver Operating Characteristic curve analysis demonstrated that PRSS22 exhibits extremely high accuracy in the diagnosis of CRC, holding great promise as a novel biomarker for CRC diagnosis.
Functional experiments confirmed that silencing PRSS22 significantly inhibits the proliferation and migration of CRC cells and induces cancer cell death. In a mouse xenograft model, knocking down PRSS22 effectively reduced tumour volume and weight, fully verifying the pro-tumorigenic function of PRSS22. Further mechanistic studies revealed that PRSS22 regulates CRC progression through three interconnected pathways: first, it inhibits the expression of the HMOX1 gene, blocking HMOX1-mediated ferroptosis, reducing reactive oxygen species accumulation and iron overload, and thus maintaining cancer cell survival; second, as a protease, it cleaves osteopontin (OPN) to generate fragments with pro-migratory activity, enhancing the invasive and metastatic capabilities of CRC cells; third, it upregulates tumour cell-derived transforming growth factor β (TGFβ), promoting the polarization of macrophages toward an M2 immunosuppressive phenotype, remodelling the tumour microenvironment, and facilitating cancer cell immune escape.
This study is the first to reveal the core regulatory role of PRSS22 in the inflammation-cancer transition of CRC, establishing a molecular network consisting of three key pathways: PRSS22-HMOX1-ferroptosis, PRSS22-OPN-cell migration and PRSS22-TGFβ-M2 macrophage polarization. It provides a new perspective for understanding the pathogenesis of CRC. Since PRSS22 is scarcely expressed in normal colorectal tissues, inhibition of PRSS22 is expected to efficiently kill cancer cells while minimizing damage to normal tissues, demonstrating excellent potential for clinical translation.
This research work was supported by multiple scientific research funds, including SCUT's Double First Class initiative, the Program for Guangdong Introducing Innovative and Entrepreneurial Teams, the Australian National Health and Medical Research Council, the Flinders Foundation, and the Shanghai Eastern Talent Program.