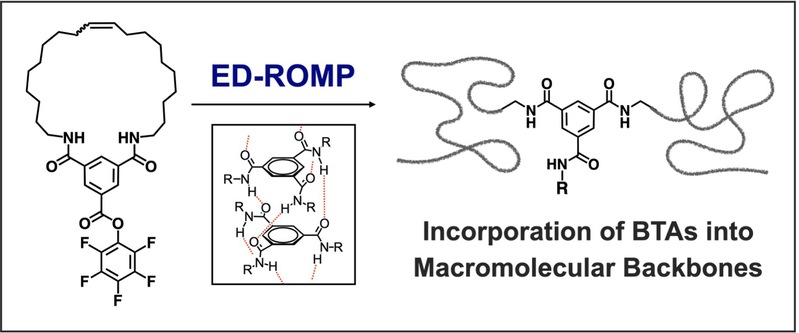
Incorporating Supramolecular Motifs into Macromolecular Backbones via Entropy-Driven Ring-Opening Metathesis Polymerization: A Case Study on Benzene-1,3,5-Tricarboxamides
Abstract:
The incorporation of supramolecular motifs into polymer architectures provides a versatile route to creating macromolecular systems with precise structures and customized properties. While numerous systems have been developed with supramolecular motifs as side chains or terminal units, methods for their controlled integration into the polymer backbone remain comparatively limited. In this work, we introduce entropy-driven ring-opening metathesis polymerization (ED-ROMP) to synthesize polymers containing the benzene-1,3,5-tricarboxamide (BTA) motif directly within the backbone. This approach was enabled by the design and synthesis of novel macrocyclic olefin monomers featuring either the BTA motif or its precursor. These monomers underwent efficient ED-ROMP, yielding a range of polymer architectures via homopolymerization, block copolymerization, and post-polymerization modification. Driven by BTA-based noncovalent interactions, the resulting polymers assembled into supramolecular nanoparticles in solution or supramolecular networks in the bulk state, demonstrating the broad potential of this synthetic strategy for constructing functional supramolecular materials.