The treatment of tumor mainly includes surgical excision, chemotherapy, radiation therapy and immunotherapy. Except for surgery, these methods treat cancer either by inhibiting tumor cell proliferation or by activating immune cell to kill tumor cell. However, the efficiency of monotherapy is not satisfying. Although researchers have proved that the combination of two or three therapies could improve the responsivity of drugs, combination therapy needs lots of experimental tests and medical costs.
Owing to these problems mentioned above, our team found that CD155 could be an appropriate target, which was proven to own a dual function on tumor cell and immune cell. On one hand, CD155 was an oncogene which could promote the proliferation and metastasis of tumor cell. On the other hand, CD155 was identified as an inhibitory immune checkpoint which could bind with TIGIT on NK and T cell to suppress the cytotoxic function of these cells. Based on previous experiments, we encapsulated siRNA targeting CD155 into a cationic lipid-assisted nanoparticle (CLANsiCD155) by double emulsion method. We demonstrated that CLAN could efficiently delivery siCD155 into tumor cell and tumor-infiltrating macrophage to downregulate CD155. CLANsiCD155 could efficiently suppress the growth of melanoma by inhibiting the proliferation of melanoma cell and activating NK and T cell via immune checkpoint function. This strategy realized the dual function by targeting one molecule. This study has recently been published in Biomaterials Science titled “Dually Regulating the Proliferation and the Immune Microenvironment of Melanoma via Nanoparticle-Delivered siRNA Targeting Onco-Immunologic CD155”.
The link of full text was showed as follow: https://pubs.rsc.org/en/content/articlehtml/2020/bm/d0bm01420f.
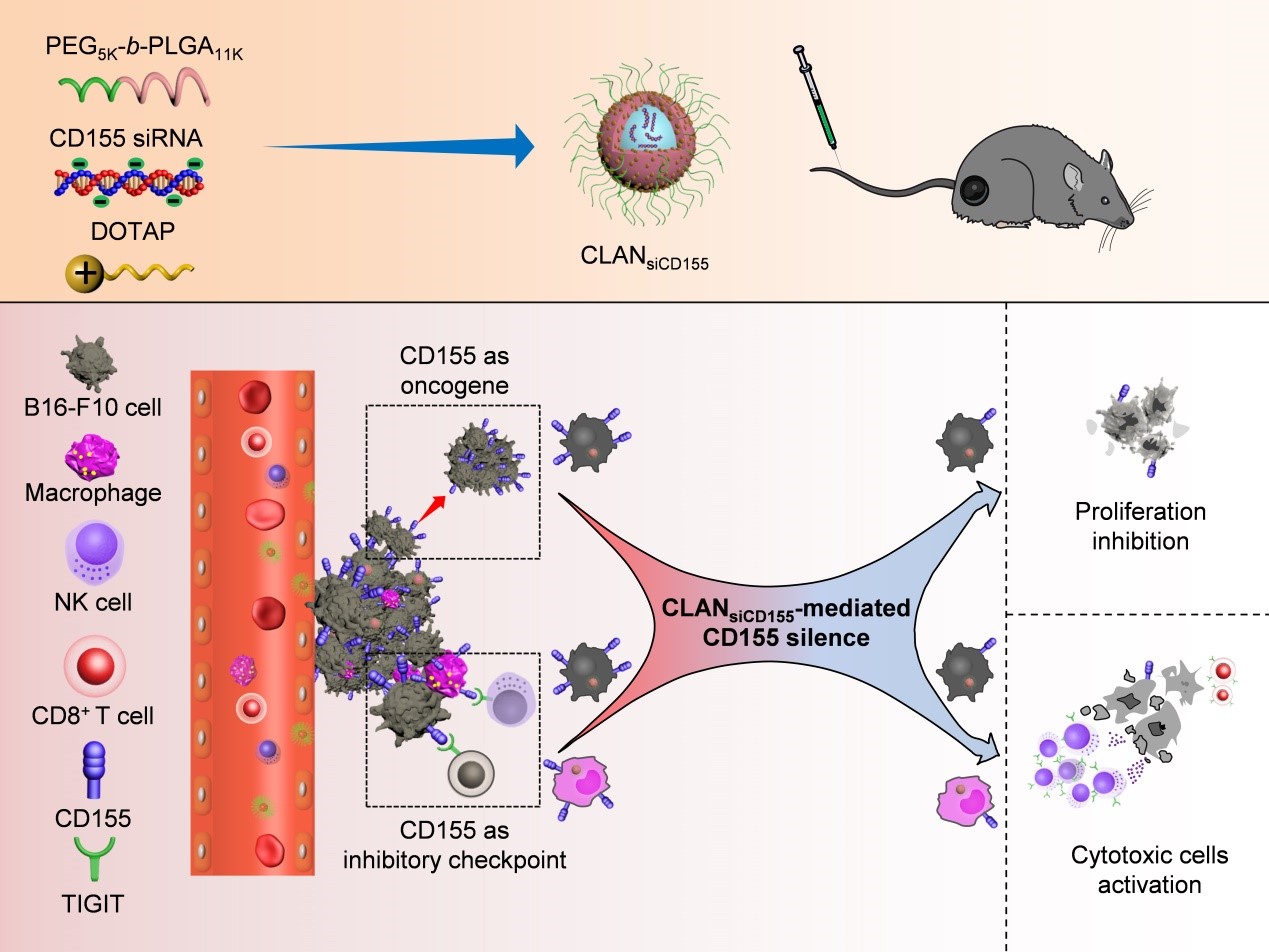
The first author of the paper is Doctoral candidate Wang Yan, and the corresponding authors are Professor Wang Jun, assistant professor Xu Congfei and post-doctor Luo Yingli. South China University of Technology is the first signature unit and the communication unit of the paper. This work was supported by the National Key R&D Program of China, the National Natural Science Foundation of China.
